A systematic review and meta-analysis of Ginsenoside-Rg1 (G-Rg1) in experimental ischemic stroke
- PMID: 25600516
- PMCID: PMC5379000
- DOI: 10.1038/srep07790
A systematic review and meta-analysis of Ginsenoside-Rg1 (G-Rg1) in experimental ischemic stroke
Abstract
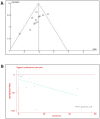
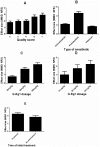
The neuroprotective actions of Ginsenoside-Rg1 (G-Rg1) have been documented for experimental stroke therapy. We used a systematic review and meta-analysis to assess the efficacy of G-Rg1 in experimental ischemic stroke. We identified studies describing the efficacy of G-Rg1 in animal models of focal cerebral ischemia. Primary outcomes were infarct volume and neurological function score (NFS). In all, eleven studies reported significant effects of G-Rg1 for improving the NFS when compared with the control group (P < 0.00001), and four studies reported significant effects of G-Rg1 for reducing infarct volume compared with middle cerebral artery occlusion group (P < 0.00001). Meanwhile, studies reported G-Rg1 was more efficacious than positive control drug nimodipine (0.7 or 1 mg/kg, intraperitoneal) according to NFS (P = 0.009) and infarct volume (p = 0.0002). The results demonstrate a marked efficacy of G-Rg1 in experimental acute ischemic stroke, but raise concerns that our value of effect size might be overestimate due to factors such as study quality and possible publication bias. Even so, the findings suggest G-Rg1 as a candidate neuroprotective drug for human ischemic stroke.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




References
-
- Kidwell C. S., Liebeskind D. S., Starkman S. & Saver J. L. Trends in acute ischemic stroke trials through the 20th century. Stroke 32, 1349–1359 (2001). - PubMed
-
- Simon R. P., Greenberg D. A. & Aminoff M. J. Clinical Neurology. 7th Edition. (McGraw-Hill Publishing House, New York, 2009).
-
- O'Collins V. E. et al. 1,026 experimental treatments in acute stroke. Ann Neurol 59, 467–477 (2006). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

