Reactive oxygen species-induced TXNIP drives fructose-mediated hepatic inflammation and lipid accumulation through NLRP3 inflammasome activation
- PMID: 25602171
- PMCID: PMC4367240
- DOI: 10.1089/ars.2014.5868
Reactive oxygen species-induced TXNIP drives fructose-mediated hepatic inflammation and lipid accumulation through NLRP3 inflammasome activation
Abstract
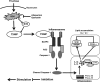
Aims: Increased fructose consumption predisposes the liver to nonalcoholic fatty liver disease (NAFLD), but the mechanisms are elusive. Thioredoxin-interacting protein (TXNIP) links oxidative stress to NOD-like receptor family, pyrin domain containing 3 (NLRP3) inflammasome activation and this signaling axis may be involved in fructose-induced NAFLD. Here, we explore the role of reactive oxygen species (ROS)-induced TXNIP overexpression in fructose-mediated hepatic NLRP3 inflammasome activation, inflammation, and lipid accumulation.
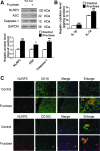
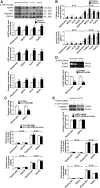
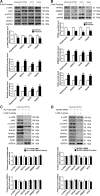
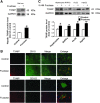
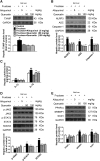
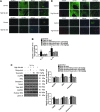
Results: Rats were fed a 10% fructose diet for 8 weeks and treated with allopurinol and quercetin during the last 4 weeks. Five millimolars of fructose-exposed hepatocytes (primary rat hepatocytes, rat hepatic parenchymal cells [RHPCs], HLO2, HepG2) were co-incubated with antioxidants or caspase-1 inhibitor or subjected to TXNIP or NLRP3 siRNA interference. Fructose induced NLRP3 inflammasome activation and pro-inflammatory cytokine secretion, janus-activated kinase 2/signal transducers and activators of transcription 3-mediated inflammatory signaling, and expression alteration of lipid metabolism-related genes in cultured hepatocytes and rat livers. NLRP3 silencing and caspase-1 suppression blocked these effects in primary rat hepatocytes and RHPCs, confirming that inflammasome activation alters hepatocyte lipid metabolism. Hepatocellular ROS and TXNIP were increased in animal and cell models. TXNIP silencing blocked NLRP3 inflammasome activation, inflammation, and lipid metabolism perturbations but not ROS induction in fructose-exposed hepatocytes, whereas antioxidants addition abrogated TXNIP induction and diminished the detrimental effects in fructose-exposed hepatocytes and rat livers.
Innovation and conclusions: This study provides a novel mechanism for fructose-induced NAFLD pathogenesis by which the ROS-TXNIP pathway mediates hepatocellular NLRP3 inflammasome activation, inflammation and lipid accumulation. Antioxidant-based interventions can inhibit the ROS-TXNIP pathway.
Figures






References
-
- Bakker PJ, Butter LM, Kors L, Teske GJ, Aten J, Sutterwala FS, Florquin S, and Leemans JC. Nlrp3 is a key modulator of diet-induced nephropathy and renal cholesterol accumulation. Kidney Int 85: 1112–1122, 2014 - PubMed
-
- Bischoff SC. Quercetin: potentials in the prevention and therapy of disease. Curr Opin Clin Nutr Metab Care 11: 733–740, 2008 - PubMed
-
- Cooper SR, Taylor JK, Miraglia LJ, and Dean NM. Pharmacology of antisense oligonucleotide inhibitors of protein expression. Pharmacol Ther 82: 427–435, 1999 - PubMed
-
- Crooke ST. Basic principles of antisense therapeutics. In: Antisense Research and Application. edited by Crooke ST. Berlin: Springer, 1998, pp. 1–50
-
- Demirel U, Yalniz M, Aygun C, Orhan C, Tuzcu M, Sahin K, Ozercan IH, and Bahcecioglu IH. Allopurinol ameliorates thioacetamide-induced acute liver failure by regulating cellular redox-sensitive transcription factors in rats. Inflammation 35: 1549–1557, 2012 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

