Phase III open-label randomized study of eribulin mesylate versus capecitabine in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline and a taxane
- PMID: 25605862
- PMCID: PMC4463422
- DOI: 10.1200/JCO.2013.52.4892
Phase III open-label randomized study of eribulin mesylate versus capecitabine in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline and a taxane
Abstract
Purpose: This phase III randomized trial (ClinicalTrials.gov identifier: NCT00337103) compared eribulin with capecitabine in patients with locally advanced or metastatic breast cancer (MBC).
Patients and methods: Women with MBC who had received prior anthracycline- and taxane-based therapy were randomly assigned to receive eribulin or capecitabine as their first-, second-, or third-line chemotherapy for advanced/metastatic disease. Stratification factors were human epidermal growth factor receptor-2 (HER2) status and geographic region. Coprimary end points were overall survival (OS) and progression-free survival (PFS).
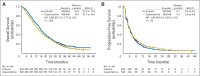
Results: Median OS times for eribulin (n = 554) and capecitabine (n = 548) were 15.9 and 14.5 months, respectively (hazard ratio [HR], 0.88; 95% CI, 0.77 to 1.00; P = .056). Median PFS times for eribulin and capecitabine were 4.1 and 4.2 months, respectively (HR, 1.08; 95% CI, 0.93 to 1.25; P = .30). Objective response rates were 11.0% for eribulin and 11.5% for capecitabine. Global health status and overall quality-of-life scores over time were similar in the treatment arms. Both treatments had manageable safety profiles consistent with their known adverse effects; most adverse events were grade 1 or 2.
Conclusion: In this phase III study, eribulin was not shown to be superior to capecitabine with regard to OS or PFS.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures

References
-
- American Cancer Society. Cancer facts and figures 2011. http://www.cancer.org/Research/CancerFactsFigures/CancerFactsFigures/can....
-
- National Cancer Institute. SEER Cancer Statistics Review, 1975-2010, National Cancer Institute. Bethesda, MD, based on November 2012 SEER data submission, posted to the SEER Web site. http://seer.cancer.gov/csr/1975_2010.
-
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology Breast Cancer Version 3. http://www.nccn.org/professionals/physician_gls/PDF/breast.pdf.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

