Autophagy in the fight against tuberculosis
- PMID: 25607549
- PMCID: PMC4390021
- DOI: 10.1089/dna.2014.2745
Autophagy in the fight against tuberculosis
Abstract
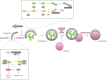
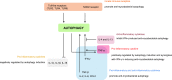
Tuberculosis (TB), a chronic infectious disease mainly caused by the tubercle bacillus Mycobacterium tuberculosis, is one of the world's deadliest diseases that has afflicted humanity since ancient times. Although the number of people falling ill with TB each year is declining, its incidence in many developing countries is still a major cause of concern. Upon invading host cells by phagocytosis, M. tuberculosis can replicate within infected cells by arresting the maturation of the phagosome whose function is to target the pathogen for elimination. Host cells have mechanisms of controlling this evasion by inducing autophagy, an elaborate cellular process that targets bacteria for progressive elimination, decreasing bacterial loads within infected cells. In addition, autophagy activation also aids in the control of inflammation, contributing to a more efficient innate immune response against M. tuberculosis. Several innovative TB therapies have been envisaged based on autophagy manipulation, with some of them revealing high potential for future clinical trials and eventual implementation in healthcare systems. Thus, this review highlights the recent advances on the innate immune response regulation by autophagy upon M. tuberculosis infection and the promising new autophagy-based therapies for TB.
Figures

References
-
- Abdallah A.M., Gey van Pittius N.C., Champion P.A., Cox J., Luirink J., Vandenbroucke-Grauls C.M., et al. (2007). Type VII secretion—mycobacteria show the way. Nat Rev Microbiol 5,883–891 - PubMed
-
- Bach H., Papavinasasundaram K.G., Wong D., Hmama Z., and Av-Gay Y. (2008). Mycobacterium tuberculosis virulence is mediated by PtpA dephosphorylation of human vacuolar protein sorting 33B. Cell Host Microbe 3,316–322 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

