Opposing regulation of endolysosomal pathways by long-acting nanoformulated antiretroviral therapy and HIV-1 in human macrophages
- PMID: 25608975
- PMCID: PMC4307176
- DOI: 10.1186/s12977-014-0133-5
Opposing regulation of endolysosomal pathways by long-acting nanoformulated antiretroviral therapy and HIV-1 in human macrophages
Abstract
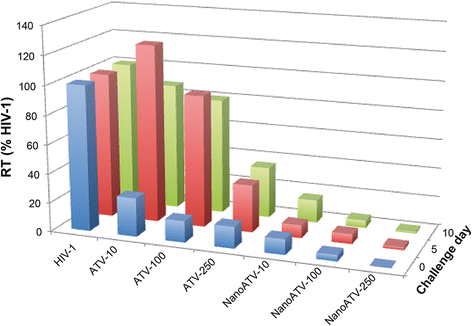
Background: Long-acting nanoformulated antiretroviral therapy (nanoART) is designed to improve patient regimen adherence, reduce systemic drug toxicities, and facilitate clearance of human immunodeficiency virus type one (HIV-1) infection. While nanoART establishes drug depots within recycling and late monocyte-macrophage endosomes, whether or not this provides a strategic advantage towards viral elimination has not been elucidated.
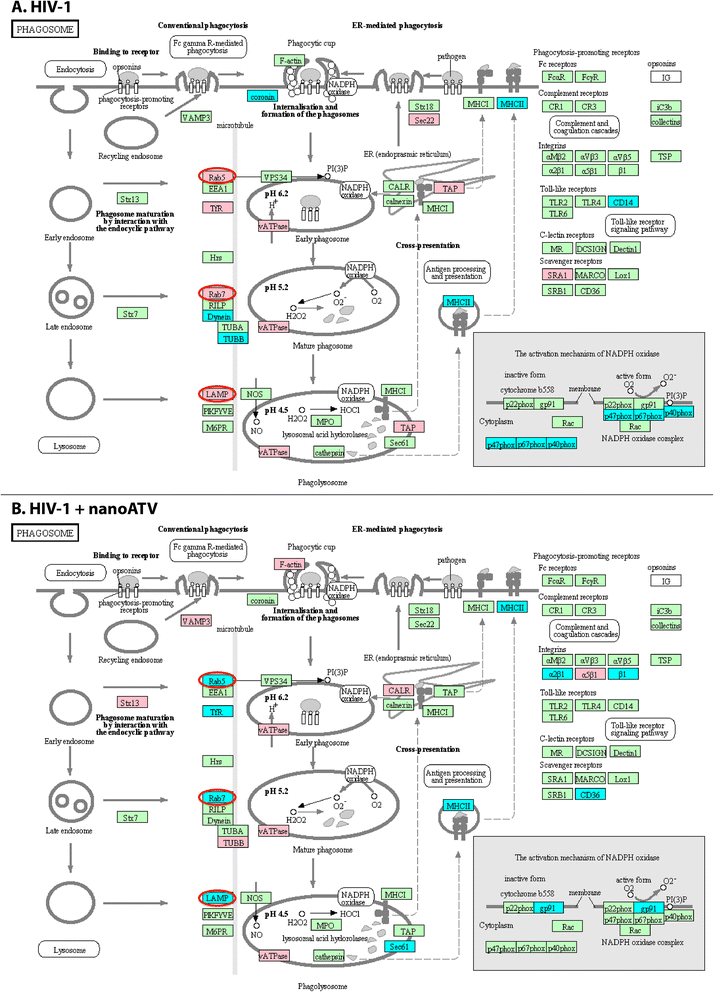
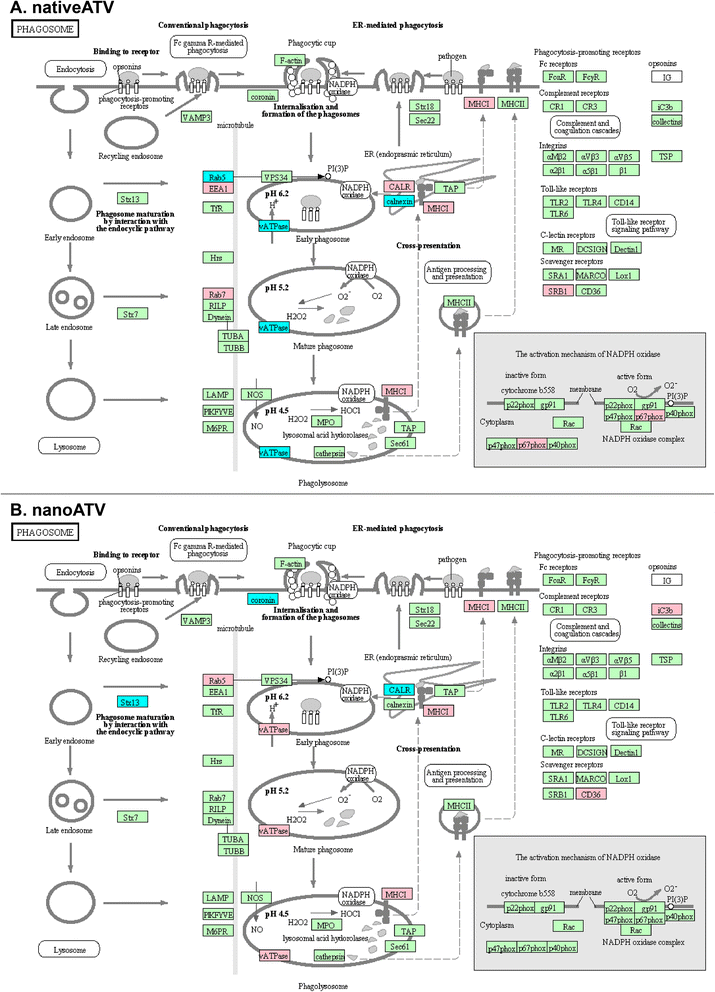
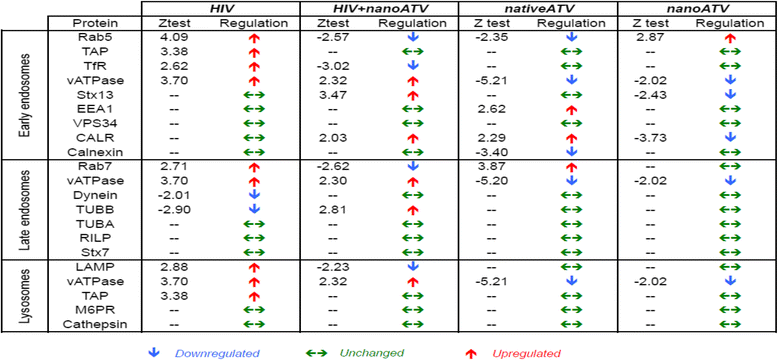
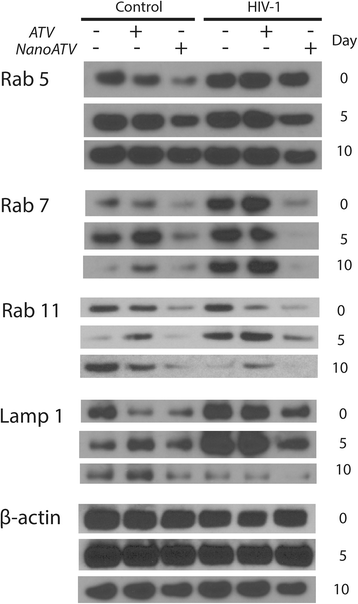
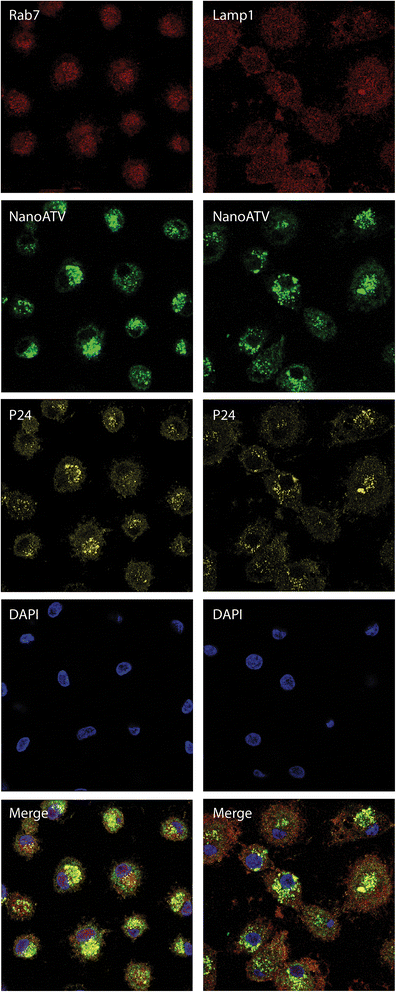
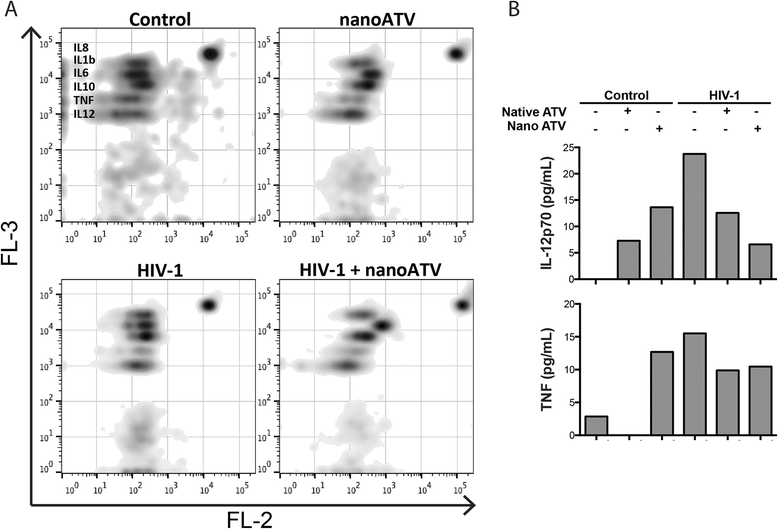
Results: We applied quantitative SWATH-MS proteomics and cell profiling to nanoparticle atazanavir (nanoATV)-treated and HIV-1 infected human monocyte-derived macrophages (MDM). Native ATV and uninfected cells served as controls. Both HIV-1 and nanoATV engaged endolysosomal trafficking for assembly and depot formation, respectively. Notably, the pathways were deregulated in opposing manners by the virus and the nanoATV, likely by viral clearance. Paired-sample z-scores, of the proteomic data sets, showed up- and down- regulation of Rab-linked endolysosomal proteins. NanoART and native ATV treated uninfected cells showed limited effects. The data was confirmed by Western blot. DAVID and KEGG bioinformatics analyses of proteomic data showed relationships between secretory, mobility and phagocytic cell functions and virus and particle trafficking.
Conclusions: We posit that modulation of endolysosomal pathways by antiretroviral nanoparticles provides a strategic path to combat HIV infection.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS036126/NS/NINDS NIH HHS/United States
- P01 NS043985/NS/NINDS NIH HHS/United States
- P01 DA028555/DA/NIDA NIH HHS/United States
- P30 MH062261/MH/NIMH NIH HHS/United States
- P01 NS031492/NS/NINDS NIH HHS/United States
- P01 NS31492/NS/NINDS NIH HHS/United States
- 2R01 NS034239/NS/NINDS NIH HHS/United States
- P01 MH64570/MH/NIMH NIH HHS/United States
- P01 MH064570/MH/NIMH NIH HHS/United States
- R01 DA030962/DA/NIDA NIH HHS/United States
- R01 AG043540/AG/NIA NIH HHS/United States
- R01 NS034239/NS/NINDS NIH HHS/United States
- P01 NS43985/NS/NINDS NIH HHS/United States
- R01 NS36126/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

