Solid microparticles based on chitosan or methyl-β-cyclodextrin: a first formulative approach to increase the nose-to-brain transport of deferoxamine mesylate
- PMID: 25620068
- PMCID: PMC4330128
- DOI: 10.1016/j.jconrel.2015.01.025
Solid microparticles based on chitosan or methyl-β-cyclodextrin: a first formulative approach to increase the nose-to-brain transport of deferoxamine mesylate
Abstract
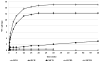
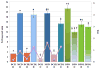
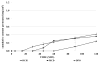
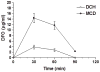
We propose the formulation and characterization of solid microparticles as nasal drug delivery systems able to increase the nose-to-brain transport of deferoxamine mesylate (DFO), a neuroprotector unable to cross the blood brain barrier and inducing negative peripheral impacts. Spherical chitosan chloride and methyl-β-cyclodextrin microparticles loaded with DFO (DCH and MCD, respectively) were obtained by spray drying. Their volume-surface diameters ranged from 1.77 ± 0.06 μm (DCH) to 3.47 ± 0.05 μm (MCD); the aerodynamic diameters were about 1.1 μm and their drug content was about 30%. In comparison with DCH, MCD enhanced the in vitro DFO permeation across lipophilic membranes, similarly as shown by ex vivo permeation studies across porcine nasal mucosa. Moreover, MCD were able to promote the DFO permeation across monolayers of PC 12 cells (neuron-like), but like DCH, it did not modify the DFO permeation pattern across Caco-2 monolayers (epithelial-like). Nasal administration to rats of 200 μg DFO encapsulated in the microparticles resulted in its uptake into the cerebrospinal fluid (CSF) with peak values ranging from 3.83 ± 0.68 μg/mL (DCH) to 14.37 ± 1.69 μg/mL (MCD) 30 min after insufflation of microparticles. No drug CSF uptake was detected after nasal administration of a DFO water solution. The DFO systemic absolute bioavailabilities obtained by DCH and MCD nasal administration were 6% and 15%, respectively. Chitosan chloride and methyl-β-cyclodextrins appear therefore suitable to formulate solid microparticles able to promote the nose to brain uptake of DFO and to limit its systemic exposure.
Keywords: Chitosan chloride; Deferoxamine mesylate; Methyl-β-cyclodextrin; Nasal formulations; Nose-to-brain transport; Pharmacokinetic studies.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures


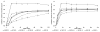

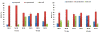

Similar articles
-
Challenges and Opportunities of Deferoxamine Delivery for Treatment of Alzheimer's Disease, Parkinson's Disease, and Intracerebral Hemorrhage.Mol Pharm. 2021 Feb 1;18(2):593-609. doi: 10.1021/acs.molpharmaceut.0c00474. Epub 2020 Oct 9. Mol Pharm. 2021. PMID: 32926630 Free PMC article. Review.
-
Cyclodextrin complexed insulin encapsulated hydrogel microparticles: An oral delivery system for insulin.J Control Release. 2010 Nov 1;147(3):377-84. doi: 10.1016/j.jconrel.2010.08.007. Epub 2010 Aug 19. J Control Release. 2010. PMID: 20727924
-
Brain targeting of resveratrol by nasal administration of chitosan-coated lipid microparticles.Eur J Pharm Biopharm. 2018 Jun;127:250-259. doi: 10.1016/j.ejpb.2018.02.010. Epub 2018 Feb 24. Eur J Pharm Biopharm. 2018. PMID: 29486302
-
Methyl-β-cyclodextrin quaternary ammonium chitosan conjugate: nanoparticles vs macromolecular soluble complex.Int J Nanomedicine. 2018 Apr 24;13:2531-2541. doi: 10.2147/IJN.S160987. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29731628 Free PMC article.
-
Nose-to-brain drug delivery: An update on clinical challenges and progress towards approval of anti-Alzheimer drugs.J Control Release. 2018 Jul 10;281:139-177. doi: 10.1016/j.jconrel.2018.05.011. Epub 2018 May 24. J Control Release. 2018. PMID: 29772289 Review.
Cited by
-
Ion-activated In Situ Gel of Gellan Gum Containing Chrysin for Nasal Administration in Parkinson's Disease.Recent Adv Drug Deliv Formul. 2024;18(1):35-49. doi: 10.2174/0126673878279656231204103855. Recent Adv Drug Deliv Formul. 2024. PMID: 38058093
-
Inhalable Antitubercular Therapy Mediated by Locust Bean Gum Microparticles.Molecules. 2016 May 28;21(6):702. doi: 10.3390/molecules21060702. Molecules. 2016. PMID: 27240337 Free PMC article.
-
Versatile Nasal Application of Cyclodextrins: Excipients and/or Actives?Pharmaceutics. 2021 Jul 30;13(8):1180. doi: 10.3390/pharmaceutics13081180. Pharmaceutics. 2021. PMID: 34452141 Free PMC article. Review.
-
Crossing the Blood-Brain Barrier: Recent Advances in Drug Delivery to the Brain.CNS Drugs. 2017 Feb;31(2):109-133. doi: 10.1007/s40263-016-0405-9. CNS Drugs. 2017. PMID: 28101766 Review.
-
Odorants could elicit repair processes in melanized neuronal and skin cells.Neural Regen Res. 2017 Sep;12(9):1401-1404. doi: 10.4103/1673-5374.215246. Neural Regen Res. 2017. PMID: 29089976 Free PMC article. Review.
References
-
- Hua Y, Keep RF, Hoff JT, Xi G. Deferoxamine therapy for intracerebral haemorrhage. Acta Neurochir Suppl. 2008;105:3–6. - PubMed
-
- [Accessed on 5 december 2014];High-Dose Deferoxamine in Intracerebral Hemorrhage (HI-DEF) Available at: https://clinicaltrials.gov/ct2/show/NCT01662895?term=deferoxamine&rank=2. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

