Sickle erythrocytes and platelets augment lung leukotriene synthesis with downregulation of anti-inflammatory proteins: relevance in the pathology of the acute chest syndrome
- PMID: 25621162
- PMCID: PMC4278608
- DOI: 10.1086/677363
Sickle erythrocytes and platelets augment lung leukotriene synthesis with downregulation of anti-inflammatory proteins: relevance in the pathology of the acute chest syndrome
Abstract
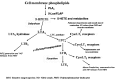
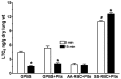
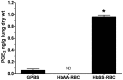
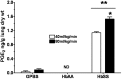
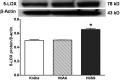
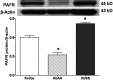
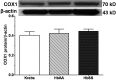
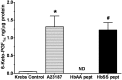
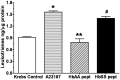
Initiation, progression, and resolution of vaso-occlusive pain episodes in sickle cell disease (SCD) have been recognized as reperfusion injury, which provokes an inflammatory response in the pulmonary circulation. Some 5-lipoxygenase (5-lox) metabolites are potent vasoconstrictors in the pulmonary circulation. We studied stimulation of production of the inflammatory eicosanoids leukotrienes (LTs) and prostaglandin E2 (PGE2) by isolated rat lungs perfused with sickle (HbSS) erythrocytes. Our hypothesis is that HbSS erythrocytes produce more LTs than normal (HbAA) erythrocytes, which can induce vaso-occlusive episodes in SCD patients. Lung perfusates were collected at specific time points and purified by high-pressure liquid chromatography, and LTC4 and PGE2 contents were measured by enzyme-linked immunosorbent assay (ELISA). Rat lung explants were also cultured with purified HbAA and HbSS peptides, and 5-lox, cyclooxygenase 1/2, and platelet-activating factor receptor (PAFR) proteins were measured by Western blotting, while prostacyclin and LTs produced by cultured lung explants were measured by ELISA. Lung weight gain and blood gas data were not different among the groups. HbSS-perfused lungs produced more LTC4 and PGE2 than HbAA-perfused lungs: 10.40 ± 0.62 versus 0.92 ± 0.2 ng/g dry lung weight (mean ± SEM; P = 0.0001) for LTC4. Inclusion of autologous platelets (platelet-rich plasma) elevated LTC4 production to 12.6 ± 0.96 and 7 ± 0.60 ng/g dry lung weight in HbSS and HbAA perfusates, respectively. HbSS lungs also expressed more 5-lox and PAFR. The data suggest that HbSS erythrocytes and activated platelets in patient's pulmonary microcirculation will enhance the synthesis and release of the proinflammatory mediators LTC4 and PGE2, both of which may contribute to onset of the acute chest syndrome in SCD.
Keywords: 5-lipoxygenase; PAF receptor; cyclooxygenase; leukotriene receptors; prostaglandins; pulmonary circulation.
Figures




Similar articles
-
Sickle erythrocytes induce prostacyclin and thromboxane synthesis by isolated perfused rat lungs.Am J Physiol. 1997 Apr;272(4 Pt 1):L597-602. doi: 10.1152/ajplung.1997.272.4.L597. Am J Physiol. 1997. PMID: 9142930
-
Elevated urinary levels of thromboxane and prostacyclin metabolities in sickle cell disease reflects activated platelets in the circulation.Br J Haematol. 1994 Jul;87(3):580-5. doi: 10.1111/j.1365-2141.1994.tb08316.x. Br J Haematol. 1994. PMID: 7993800
-
Significance of endothelial prostacyclin and nitric oxide in peripheral and pulmonary circulation.Med Sci Monit. 2001 Jan-Feb;7(1):1-16. Med Sci Monit. 2001. PMID: 11208485
-
Leukotrienes: possible mediators in bronchial asthma.Eur J Respir Dis Suppl. 1983;129:45-64. Eur J Respir Dis Suppl. 1983. PMID: 6317422 Review.
-
Leukotriene pathway in sickle cell disease: a potential target for directed therapy.Expert Rev Hematol. 2009 Feb;2(1):57-68. doi: 10.1586/17474086.2.1.57. Expert Rev Hematol. 2009. PMID: 21082995 Review.
Cited by
-
Inflammatory targets of therapy in sickle cell disease.Transl Res. 2016 Jan;167(1):281-97. doi: 10.1016/j.trsl.2015.07.001. Epub 2015 Jul 11. Transl Res. 2016. PMID: 26226206 Free PMC article. Review.
-
Thrombo-Inflammation in COVID-19 and Sickle Cell Disease: Two Faces of the Same Coin.Biomedicines. 2023 Jan 25;11(2):338. doi: 10.3390/biomedicines11020338. Biomedicines. 2023. PMID: 36830874 Free PMC article. Review.
-
Therapeutic strategies for sickle cell disease: towards a multi-agent approach.Nat Rev Drug Discov. 2019 Feb;18(2):139-158. doi: 10.1038/s41573-018-0003-2. Nat Rev Drug Discov. 2019. PMID: 30514970 Free PMC article. Review.
-
Hypoxia-mediated impaired erythrocyte Lands' Cycle is pathogenic for sickle cell disease.Sci Rep. 2016 Jul 20;6:29637. doi: 10.1038/srep29637. Sci Rep. 2016. PMID: 27436223 Free PMC article.
-
Airway Inflammation and Lung Function in Sickle Cell Disease.Pediatr Allergy Immunol Pulmonol. 2019 Sep 1;32(3):92-102. doi: 10.1089/ped.2019.1014. Epub 2019 Sep 17. Pediatr Allergy Immunol Pulmonol. 2019. PMID: 31559108 Free PMC article.
References
-
- Samuelsson B, Dahlen SE, Lindgren JA, Rouzer CA, Serhan CN. Leukotrienes and lipoxins: structure, biosynthesis, and biological effects. Science 1987;237(4819):1171–1176. - PubMed
-
- Voelkel NF. Species variation in the pulmonary responses to arachidonic acid metabolites. Protaglandins 1985;29(5):867–889. - PubMed
-
- Morganroth ML, Stenmark KR, Zirrolli JA, Mauldin R, Mathias M, Reeves JT, Murphy RC, Voelkel NF. Leukotriene C4 production during hypoxic vasoconstriction in the isolated rat lung. Protaglandins 1984;28(6):867–875. - PubMed
-
- Knight-Perry J, DeBaun MR, Strunk RC, Field JJ. Leukotriene pathway in sickle cell disease: a potential target for directed therapy. Expert Rev Hematol 2009;2(1):57–68. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

