Intermediate filament mechanics in vitro and in the cell: from coiled coils to filaments, fibers and networks
- PMID: 25621895
- PMCID: PMC4355244
- DOI: 10.1016/j.ceb.2015.01.001
Intermediate filament mechanics in vitro and in the cell: from coiled coils to filaments, fibers and networks
Abstract
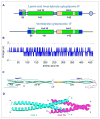
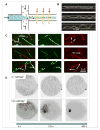
Intermediate filament proteins form filaments, fibers and networks both in the cytoplasm and the nucleus of metazoan cells. Their general structural building plan accommodates highly varying amino acid sequences to yield extended dimeric α-helical coiled coils of highly conserved design. These 'rod' particles are the basic building blocks of intrinsically flexible, filamentous structures that are able to resist high mechanical stresses, that is, bending and stretching to a considerable degree, both in vitro and in the cell. Biophysical and computer modeling studies are beginning to unfold detailed structural and mechanical insights into these major supramolecular assemblies of cell architecture, not only in the 'test tube' but also in the cellular and tissue context.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures

References
-
- Parry DAD, Steinert M. Intermediate filaments: molecular architecture, assembly, dynamics and polymorphism. Quart Rev Biophys. 1999;32:99–187. - PubMed
-
- Erber A, Riemer D, Hofemeister H, Bovenschulte M, Stick R, Panopoulou G, Lehrach H, Weber K. Characterization of the Hydra lamin and its gene: A molecular phylogeny of metazoan lamins. J Mol Evol. 1999;49:260–271. - PubMed
-
- Herrmann H, Aebi U. Intermediate Filaments: Molecular Structure, Assembly Mechanism, and Integration Into Functionally Distinct Intracellular Scaffolds. Ann Rev Biochem. 2004;73:749–789. - PubMed
-
- Herrmann H, Kreplak L, Aebi U. Isolation, characterization, and in vitro assembly of intermediate filaments. Methods Cell Biol. 2004;78:3–24. - PubMed
-
- Herrmann H, Häner M, Brettel M, Müller SA, Goldie KN, Fedtke B, Lustig A, Franke WW, Aebi U. Structure and Assembly Properties of the Intermediate Filament Protein Vimentin: The Role of its Head, Rod and Tail Domains. J Mol Biol. 1996;264:933–953. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

