Lentivirus-mediated RNAi knockdown of the gap junction protein, Cx43, attenuates the development of vascular restenosis following balloon injury
- PMID: 25625334
- PMCID: PMC4356439
- DOI: 10.3892/ijmm.2015.2078
Lentivirus-mediated RNAi knockdown of the gap junction protein, Cx43, attenuates the development of vascular restenosis following balloon injury
Abstract
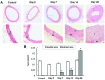
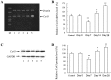
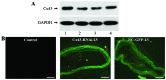
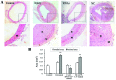
Percutaneous coronary intervention [PCI or percutaneous transluminal coronary angioplasty (PTCA)] has been developed into a mature interventional treatment for atherosclerotic cardiovascular disease. However, the long-term therapeutic effect is compromised by the high incidence of vascular restenosis following angioplasty, and the underlying mechanisms of vascular restenosis have not yet been fully elucidated. In the present study, we investigated the role of the gap junction (GJ) protein, connexin 43 (Cx43), in the development of vascular restenosis. To establish vascular restenosis, rat carotid arteries were subjected to balloon angioplasty injury. At 0, 7, 14 and 2 days following balloon injury, the arteries were removed, and the intimal/medial area of the vessels was measured to evaluate the degree of restenosis. We found that the intimal area gradually increased following balloon injury. Intimal hyperplasia and restenosis were particularly evident at 14 and 28 days after injury. In addition, the mRNA and protein expression of Cx43 was temporarily decreased at 7 days, and subsequently increased at 14 and 28 days following balloon injury, as shown by RT-PCR and western blot analysis. To determine the involvement of Cx43 in vascular restenosis, the lentivirus vector expressing shRNA targeting Cx43, Cx43-RNAi-LV, was used to silence Cx43 in the rat carotid arteries. The knockdown of Cx43 effectively attenuated the development of intimal hyperplasia and vascular restenosis following balloon injury. Thus, our data indicate the vital role of the GJ protein, Cx43, in the development of vascular restenosis, and provide new insight into the pathogenesis of vascular restenosis. Cx43 may prove to be a novel potential pharmacological target for the prevention of vascular restenosis following PCI.
Figures

Similar articles
-
Knockdown of connexin 43 attenuates balloon injury-induced vascular restenosis through the inhibition of the proliferation and migration of vascular smooth muscle cells.Int J Mol Med. 2015 Nov;36(5):1361-8. doi: 10.3892/ijmm.2015.2346. Epub 2015 Sep 15. Int J Mol Med. 2015. PMID: 26398676
-
Reduced connexin43 expression limits neointima formation after balloon distension injury in hypercholesterolemic mice.Circulation. 2006 Jun 20;113(24):2835-43. doi: 10.1161/CIRCULATIONAHA.106.627703. Epub 2006 Jun 12. Circulation. 2006. PMID: 16769907
-
Lentivirus-mediated RNAi targeting CREB binding protein attenuates neointimal formation and promotes re-endothelialization in balloon injured rat carotid artery.Cell Physiol Biochem. 2010;26(3):441-8. doi: 10.1159/000320567. Epub 2010 Aug 24. Cell Physiol Biochem. 2010. PMID: 20798529
-
[Preventing effect of angiotensin II receptor blocker on neointimal hyperplasia after vascular injury].Nihon Rinsho. 2002 Oct;60(10):1957-61. Nihon Rinsho. 2002. PMID: 12397691 Review. Japanese.
-
Pathophysiology of coronary artery restenosis.Rev Cardiovasc Med. 2002;3 Suppl 5:S4-9. Rev Cardiovasc Med. 2002. PMID: 12478229 Review.
Cited by
-
Experimental Liver Cirrhosis Inhibits Restenosis after Balloon Angioplasty.Int J Mol Sci. 2023 Jul 12;24(14):11351. doi: 10.3390/ijms241411351. Int J Mol Sci. 2023. PMID: 37511114 Free PMC article.
-
The effect of Telmisartan on the expression of connexin43 and neointimal hyperplasia in a rabbit iliac artery restenosis model.Heart Vessels. 2019 Jul;34(7):1230-1239. doi: 10.1007/s00380-018-01338-1. Epub 2019 Jan 22. Heart Vessels. 2019. PMID: 30671641
-
The role of gap junctions in cell death and neuromodulation in the retina.Neural Regen Res. 2021 Oct;16(10):1911-1920. doi: 10.4103/1673-5374.308069. Neural Regen Res. 2021. PMID: 33642359 Free PMC article. Review.
-
High expression of ubiquitin-specific peptidase 39 is associated with the development of vascular remodeling.Mol Med Rep. 2017 May;15(5):2567-2573. doi: 10.3892/mmr.2017.6297. Epub 2017 Mar 8. Mol Med Rep. 2017. PMID: 28447728 Free PMC article.
-
Yiqihuoxuejiedu Formula Restrains Vascular Remodeling by Reducing the Inflammation Reaction and Cx43 Expression in the Adventitia after Balloon Injury.Evid Based Complement Alternat Med. 2015;2015:904273. doi: 10.1155/2015/904273. Epub 2015 Oct 19. Evid Based Complement Alternat Med. 2015. PMID: 26557868 Free PMC article.
References
-
- Meads C, Cummins C, Jolly K, Stevens A, Burls A, Hyde C. Coronary artery stents in the treatment of ischaemic heart disease: a rapid and systematic review. Health Technol Assess. 2000;4:1–153. - PubMed
-
- Owens GK. Regulation of differentiation of vascular smooth muscle cells. Physiol Rev. 1995;75:487–517. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

