Paraoxonase 2 serves a proapopotic function in mouse and human cells in response to the Pseudomonas aeruginosa quorum-sensing molecule N-(3-Oxododecanoyl)-homoserine lactone
- PMID: 25627690
- PMCID: PMC4358143
- DOI: 10.1074/jbc.M114.620039
Paraoxonase 2 serves a proapopotic function in mouse and human cells in response to the Pseudomonas aeruginosa quorum-sensing molecule N-(3-Oxododecanoyl)-homoserine lactone
Abstract
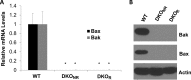
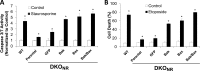
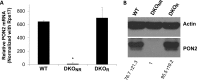
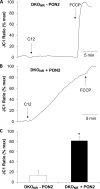
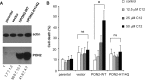
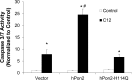
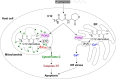
Pseudomonas aeruginosa use quorum-sensing molecules, including N-(3-oxododecanoyl)-homoserine lactone (C12), for intercellular communication. C12 activated apoptosis in mouse embryo fibroblasts (MEF) from both wild type (WT) and Bax/Bak double knock-out mice (WT MEF and DKO MEF that were responsive to C12, DKOR MEF): nuclei fragmented; mitochondrial membrane potential (Δψmito) depolarized; Ca(2+) was released from the endoplasmic reticulum (ER), increasing cytosolic [Ca(2+)] (Cacyto); and caspase 3/7 was activated. DKOR MEF had been isolated from a nonclonal pool of DKO MEF that were non-responsive to C12 (DKONR MEF). RNAseq analysis, quantitative PCR, and Western blots showed that WT and DKOR MEF both expressed genes associated with cancer, including paraoxonase 2 (PON2), whereas DKONR MEF expressed little PON2. Adenovirus-mediated expression of human PON2 in DKONR MEF rendered them responsive to C12: Δψmito depolarized, Cacyto increased, and caspase 3/7 activated. Human embryonic kidney 293T (HEK293T) cells expressed low levels of endogenous PON2, and these cells were also less responsive to C12. Overexpression of PON2, but not PON2-H114Q (no lactonase activity) in HEK293T cells caused them to become sensitive to C12. Because [C12] may reach high levels in biofilms in lungs of cystic fibrosis (CF) patients, PON2 lactonase activity may control Δψmito, Ca(2+) release from the ER, and apoptosis in CF airway epithelia. Coupled with previous data, these results also indicate that PON2 uses its lactonase activity to prevent Bax- and Bak-dependent apoptosis in response to common proapoptotic drugs like doxorubicin and staurosporine, but activates Bax- and Bak-independent apoptosis in response to C12.
Keywords: Apoptosis; Bax; Cancer; Paraoxonase 2; Pseudomonas aeruginosa (P. aeruginosa); Quorum Sensing.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures





Similar articles
-
Pseudomonas aeruginosa homoserine lactone triggers apoptosis and Bak/Bax-independent release of mitochondrial cytochrome C in fibroblasts.Cell Microbiol. 2014 Jul;16(7):1094-104. doi: 10.1111/cmi.12263. Epub 2014 Feb 13. Cell Microbiol. 2014. PMID: 24438098 Free PMC article.
-
PON2 mediates mitochondrial dysfunction in tracheal epithelial cells in response to a quorum sensing molecule N-(-3-oxododecanoyl)-l-homoserine lactone.Biochem J. 2022 Oct 14;479(19):2013-2034. doi: 10.1042/BCJ20220100. Biochem J. 2022. PMID: 36094147
-
Paraoxonase-2 deficiency enhances Pseudomonas aeruginosa quorum sensing in murine tracheal epithelia.Am J Physiol Lung Cell Mol Physiol. 2007 Apr;292(4):L852-60. doi: 10.1152/ajplung.00370.2006. Epub 2006 Nov 22. Am J Physiol Lung Cell Mol Physiol. 2007. PMID: 17122353
-
Quorum sensing molecule N-(3-oxododecanoyl)-l-homoserine lactone: An all-rounder in mammalian cell modification.J Oral Biosci. 2020 Mar;62(1):16-29. doi: 10.1016/j.job.2020.01.001. Epub 2020 Jan 23. J Oral Biosci. 2020. PMID: 31982630 Review.
-
Bacteria-Host Crosstalk: Sensing of the Quorum in the Context of Pseudomonas aeruginosa Infections.J Innate Immun. 2019;11(3):263-279. doi: 10.1159/000494069. Epub 2018 Nov 14. J Innate Immun. 2019. PMID: 30428481 Free PMC article. Review.
Cited by
-
Pseudomonas aeruginosa quorum-sensing molecule N-(3-oxododecanoyl)-L-homoserine lactone mediates Ca+2 dysregulation, mitochondrial dysfunction, and apoptosis in human peripheral blood lymphocytes.Heliyon. 2023 Oct 28;9(11):e21462. doi: 10.1016/j.heliyon.2023.e21462. eCollection 2023 Nov. Heliyon. 2023. PMID: 38027911 Free PMC article.
-
Impact of quorum sensing signaling molecules in gram-negative bacteria on host cells: current understanding and future perspectives.Gut Microbes. 2022 Jan-Dec;14(1):2039048. doi: 10.1080/19490976.2022.2039048. Gut Microbes. 2022. PMID: 35188058 Free PMC article. Review.
-
Challenges and Limitations of Anti-quorum Sensing Therapies.Front Microbiol. 2019 Oct 31;10:2473. doi: 10.3389/fmicb.2019.02473. eCollection 2019. Front Microbiol. 2019. PMID: 31736912 Free PMC article.
-
Pseudomonas aeruginosa quorum-sensing molecule N-(3-oxo-dodecanoyl)-L-homoserine lactone triggers mitochondrial dysfunction and apoptosis in neutrophils through calcium signaling.Med Microbiol Immunol. 2019 Dec;208(6):855-868. doi: 10.1007/s00430-019-00631-8. Epub 2019 Aug 3. Med Microbiol Immunol. 2019. PMID: 31377870
-
Role of paraoxonase 2 (PON2) as a potential biomarker and therapeutic target in cancer treatment.J Cancer Res Clin Oncol. 2025 Aug 12;151(8):229. doi: 10.1007/s00432-025-06282-y. J Cancer Res Clin Oncol. 2025. PMID: 40794328 Free PMC article. Review.
References
-
- Shiner E. K., Rumbaugh K. P., Williams S. C. (2005) Inter-kingdom signaling: deciphering the language of acyl homoserine lactones. FEMS Microbiol Rev. 29, 935–947 - PubMed
-
- Rumbaugh K. P., Griswold J. A., Hamood A. N. (2000) The role of quorum sensing in the in vivo virulence of Pseudomonas aeruginosa. Microbes Infect. 2, 1721–1731 - PubMed
-
- Bjarnsholt T., Ciofu O., Molin S., Givskov M., Høiby N. (2013) Applying insights from biofilm biology to drug development: can a new approach be developed? Nat. Rev. Drug Discov. 12, 791–808 - PubMed
-
- Staudinger B. J., Muller J. F., Halldórsson S., Boles B., Angermeyer A., Nguyen D., Rosen H., Baldursson O., Gottfreðsson M., Guðmundsson G. H., Singh P. K. (2014) Conditions associated with the cystic fibrosis defect promote chronic Pseudomonas aeruginosa infection. Am. J. Respir. Crit. Care Med. 189, 812–824 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

