A transendocytosis model of CTLA-4 function predicts its suppressive behavior on regulatory T cells
- PMID: 25632005
- PMCID: PMC4522736
- DOI: 10.4049/jimmunol.1401876
A transendocytosis model of CTLA-4 function predicts its suppressive behavior on regulatory T cells
Abstract
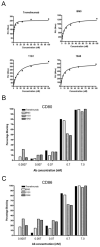
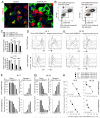
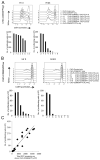
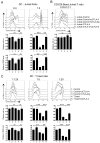
Manipulation of the CD28/CTLA-4 pathway is at the heart of a number of immunomodulatory approaches used in both autoimmunity and cancer. Although it is clear that CTLA-4 is a critical regulator of T cell responses, the immunological contexts in which CTLA-4 controls immune responses are not well defined. In this study, we show that whereas CD80/CD86-dependent activation of resting human T cells caused extensive T cell proliferation and robust CTLA-4 expression, in this context CTLA-4 blocking Abs had no impact on the response. In contrast, in settings where CTLA-4(+) cells were present as "regulators," inhibition of resting T cell responses was dependent on CTLA-4 expression and specifically related to the number of APC. At low numbers of APC or low levels of ligand, CTLA-4-dependent suppression was highly effective whereas at higher APC numbers or high levels of ligand, inhibition was lost. Accordingly, the degree of suppression correlated with the level of CD86 expression remaining on the APC. These data reveal clear rules for the inhibitory function of CTLA-4 on regulatory T cells, which are predicted by its ability to remove ligands from APC.
Copyright © 2015 by The American Association of Immunologists, Inc.
Figures

Similar articles
-
Targeting CD28, CTLA-4 and PD-L1 costimulation differentially controls immune synapses and function of human regulatory and conventional T-cells.PLoS One. 2013 Dec 23;8(12):e83139. doi: 10.1371/journal.pone.0083139. eCollection 2013. PLoS One. 2013. PMID: 24376655 Free PMC article.
-
Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4.Science. 2011 Apr 29;332(6029):600-3. doi: 10.1126/science.1202947. Epub 2011 Apr 7. Science. 2011. PMID: 21474713 Free PMC article.
-
CD86 Is a Selective CD28 Ligand Supporting FoxP3+ Regulatory T Cell Homeostasis in the Presence of High Levels of CTLA-4.Front Immunol. 2020 Dec 8;11:600000. doi: 10.3389/fimmu.2020.600000. eCollection 2020. Front Immunol. 2020. PMID: 33363541 Free PMC article.
-
Targeting T cell costimulation in autoimmune disease.Expert Opin Ther Targets. 2002 Jun;6(3):275-89. doi: 10.1517/14728222.6.3.275. Expert Opin Ther Targets. 2002. PMID: 12223069 Review.
-
A transendocytosis perspective on the CD28/CTLA-4 pathway.Adv Immunol. 2014;124:95-136. doi: 10.1016/B978-0-12-800147-9.00004-2. Adv Immunol. 2014. PMID: 25175774 Review.
Cited by
-
Versatile and High-throughput Force Measurement Platform for Dorsal Cell Mechanics.Sci Rep. 2019 Sep 16;9(1):13286. doi: 10.1038/s41598-019-49592-1. Sci Rep. 2019. PMID: 31527594 Free PMC article.
-
Translational Approaches Targeting Ceramide Generation From Sphingomyelin in T Cells to Modulate Immunity in Humans.Front Immunol. 2019 Oct 11;10:2363. doi: 10.3389/fimmu.2019.02363. eCollection 2019. Front Immunol. 2019. PMID: 31681273 Free PMC article. Review.
-
Recent Advances and Challenges in Cancer Immunotherapy.Cancers (Basel). 2022 Aug 17;14(16):3972. doi: 10.3390/cancers14163972. Cancers (Basel). 2022. PMID: 36010965 Free PMC article. Review.
-
Functional Relevance of CTLA4 Variants: an Upgraded Approach to Assess CTLA4-Dependent Transendocytosis by Flow Cytometry.J Clin Immunol. 2023 Nov;43(8):2076-2089. doi: 10.1007/s10875-023-01582-9. Epub 2023 Sep 23. J Clin Immunol. 2023. PMID: 37740092 Free PMC article.
-
Expanding the molecular and phenotypic spectrum of CTLA-4 insufficiency.Pediatr Allergy Immunol. 2024 Feb;35(2):e14077. doi: 10.1111/pai.14077. Pediatr Allergy Immunol. 2024. PMID: 38351878 Free PMC article. No abstract available.
References
-
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. - PubMed
-
- Reis e S. Dendritic cells as sensors of infection. Immunity. 2001;14:495–498. - PubMed
-
- Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat.Immunol. 2001;2:675–680. - PubMed
-
- Azuma M, Ito D, Yagita H, Okumura K, Phillips JH, Lanier LL, Somoza C. B70 antigen is a second ligand for CTLA-4 and CD28. Nature. 1993;366:76–79. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

