cFLIP is critical for oligodendrocyte protection from inflammation
- PMID: 25633192
- PMCID: PMC4532774
- DOI: 10.1038/cdd.2014.237
cFLIP is critical for oligodendrocyte protection from inflammation
Abstract
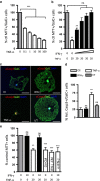
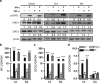
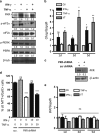
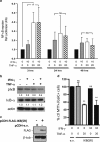
Neuroinflammation associated with degenerative central nervous system disease and injury frequently results in oligodendrocyte death. While promoting oligodendrocyte viability is a major therapeutic goal, little is known about protective signaling strategies. We report that in highly purified rat oligodendrocytes, interferon gamma (IFNγ) activates a signaling pathway that protects these cells from tumor necrosis factor alpha (TNFα)-induced cytotoxicity. IFNγ protection requires Jak (Janus kinase) activation, components of the integrated stress response and NF-κB activation. Although NF-κB activation also occurred transiently in the absence of IFNγ and presence of TNFα, this activation was not sufficient to prevent induction of the TNFα-responsive cell death pathway. Genetic inhibition of NF-κB translocation to the nucleus abrogated IFNγ-mediated protection and did not change the cell death induced by TNFα, suggesting that NF-κB activation via IFNγ induces a different set of responses than activation of NF-κB via TNFα. A promising candidate is the NF-κB target cFLIP (cellular FLICE (FADD-like IL-1β-converting enzyme)-inhibitory protein), which is protease-deficient caspase homolog that inhibits caspase-3 activation. We show that IFNγ-mediated protection led to upregulation of cFLIP. Overexpression of cFLIP was sufficient for oligodendrocyte protection from TNFα and short hairpin RNA knockdown of cFLIP-abrogated IFNγ -mediated protection. To determine the relevance of our in vitro finding to the more complex in vivo situation, we determined the impact on oligodendrocyte death of regional cFLIP loss of function in a murine model of neuroinflammation. Our data show that downregulation of cFLIP during inflammation leads to death of oligodendrocytes and decrease of myelin in vivo. Taken together, we show that IFNγ-mediated induction of cFLIP expression provides a new mechanism by which this cytokine can protect oligodendrocytes from TNFα-induced cell death.
Figures



References
-
- Agresti C, D'Urso D, Levi G. Reversible inhibitory effects of interferon-gamma and tumour necrosis factor-alpha on oligodendroglial lineage cell proliferation and differentiation in vitro. Eur J Neurosci. 1996;8:1106–1116. - PubMed
-
- Andrews T, Zhang P, Bhat NR. TNFalpha potentiates IFNgamma-induced cell death in oligodendrocyte progenitors. J Neurosci Res. 1998;54:574–583. - PubMed
-
- Baerwald KD, Popko B. Developing and mature oligodendrocytes respond differently to the immune cytokine interferon-gamma. J Neurosci Res. 1998;52:230–239. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

