Quality of reporting of randomised controlled trials of herbal interventions in ASEAN Plus Six Countries: a systematic review
- PMID: 25633206
- PMCID: PMC4310614
- DOI: 10.1371/journal.pone.0108681
Quality of reporting of randomised controlled trials of herbal interventions in ASEAN Plus Six Countries: a systematic review
Abstract
Background: Many randomised controlled trials (RCTs) of herbal interventions have been conducted in the ASEAN Communities. Good quality reporting of RCTs is essential for assessing clinical significance. Given the importance ASEAN placed on herbal medicines, the reporting quality of RCTs of herbal interventions among the ASEAN Communities deserved a special attention.
Objectives: To systematically review the quality of reporting of RCTs of herbal interventions conducted in the ASEAN Plus Six Countries.
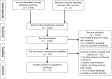
Methods: Searches were performed using PubMed, EMBASE, The Cochrane Library, and Allied and Complementary Medicine (AMED), from inception through October 2013. These were limited to studies specific to humans and RCTs. Herbal species search terms were based on those listed in the National List of Essential Medicines [NLEM (Thailand, 2011)]. Studies conducted in the ASEAN Plus Six Countries, published in English were included.
Results: Seventy-one articles were identified. Thirty (42.25%) RCTs were from ASEAN Countries, whereas 41 RCTs (57.75%) were from Plus Six Group. Adherence to the recommended CONSORT checklist items for reporting of RCTs of herbal interventions among ASEAN Plus Six Countries ranged from 0% to 97.18%. Less than a quarter of the RCTs (18.31%) reported information on standardisation of the herbal products. However, the scope of our interventions of interest was limited to those developed from 20 herbal species listed in the NLEM of Thailand.
Conclusions: The present study highlights the need to improve reporting quality of RCTs of herbal interventions across ASEAN Plus Six Communities.
Conflict of interest statement
Figures
Similar articles
-
Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals.Cochrane Database Syst Rev. 2012 Nov 14;11(11):MR000030. doi: 10.1002/14651858.MR000030.pub2. Cochrane Database Syst Rev. 2012. PMID: 23152285 Free PMC article.
-
Reporting quality and evidence support in randomized controlled trials of herbal medicine formulas for vestibular migraine.Phytomedicine. 2025 Jul 25;143:156864. doi: 10.1016/j.phymed.2025.156864. Epub 2025 May 16. Phytomedicine. 2025. PMID: 40449448
-
Herbal and dietary therapies for primary and secondary dysmenorrhoea.Cochrane Database Syst Rev. 2001;(3):CD002124. doi: 10.1002/14651858.CD002124. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2016 Mar 22;3:CD002124. doi: 10.1002/14651858.CD002124.pub2. PMID: 11687013 Updated.
-
Individual-level interventions to reduce personal exposure to outdoor air pollution and their effects on people with long-term respiratory conditions.Cochrane Database Syst Rev. 2021 Aug 9;8(8):CD013441. doi: 10.1002/14651858.CD013441.pub2. Cochrane Database Syst Rev. 2021. PMID: 34368949 Free PMC article.
-
Herbal medicines for viral myocarditis.Cochrane Database Syst Rev. 2013 Aug 28;(8):CD003711. doi: 10.1002/14651858.CD003711.pub5. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2016 May 03;(5):CD003711. doi: 10.1002/14651858.CD003711.pub6. PMID: 23986406 Updated.
Cited by
-
The critical appraisal of randomized controlled trials published in an Indian journal to assess the quality of reporting: A retrospective, cross-sectional study.Perspect Clin Res. 2022 Jan-Mar;13(1):33-37. doi: 10.4103/picr.PICR_169_19. Epub 2021 Jan 8. Perspect Clin Res. 2022. PMID: 35198426 Free PMC article.
-
The quality of reporting of randomized controlled trials of HuatuoZaizao pill for stroke.Front Pharmacol. 2023 Jan 10;13:1106957. doi: 10.3389/fphar.2022.1106957. eCollection 2022. Front Pharmacol. 2023. PMID: 36703753 Free PMC article.
-
Reporting quality of stepped wedge design randomized trials: a systematic review protocol.Clin Epidemiol. 2016 Jul 8;8:261-6. doi: 10.2147/CLEP.S103098. eCollection 2016. Clin Epidemiol. 2016. PMID: 27468249 Free PMC article.
-
In Vitro Responses of Plant Growth Factors on Growth, Yield, Phenolics Content and Antioxidant Activities of Clinacanthus nutans (Sabah Snake Grass).Plants (Basel). 2020 Aug 14;9(8):1030. doi: 10.3390/plants9081030. Plants (Basel). 2020. PMID: 32823824 Free PMC article.
-
Does the medical literature remain inadequately described despite having reporting guidelines for 21 years? - A systematic review of reviews: an update.J Multidiscip Healthc. 2018 Sep 27;11:495-510. doi: 10.2147/JMDH.S155103. eCollection 2018. J Multidiscip Healthc. 2018. PMID: 30310289 Free PMC article. Review.
References
-
- ASEAN Secretariat (2009) ASEAN Socio-Cultural Community Blueprint. ASEAN Secretariat. Available: http://www.asean.org/archive/5187-19.pdf. Accessed 9 December 2013.
-
- Urata S (2008) An ASEAN+6 Economic Partnership: Significance and Tasks. Asia Research Report 2007. Japan Center for Economic Research. Available: http://www.jcer.or.jp/eng/pdf/asia07.pdf. Accessed 10 December 2013.
-
- Linde K, Jonas WB, Melchart D, Willich S (2001) The methodological quality of randomized controlled trials of homeopathy, herbal medicines and acupuncture. International journal of epidemiology 30(3): 526–531. - PubMed
-
- Gagnier JJ, DeMelo J, Boon H, Rochon P, Bombardier C (2006) Quality of reporting of randomized controlled trials of herbal medicine interventions. The American journal of medicine 119(9): 800 e1–11. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical