Epigenetic regulation of neuronal dendrite and dendritic spine development
- PMID: 25635180
- PMCID: PMC4307848
- DOI: 10.1007/s11515-010-0650-0
Epigenetic regulation of neuronal dendrite and dendritic spine development
Abstract
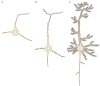
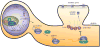
Dendrites and the dendritic spines of neurons play key roles in the connectivity of the brain and have been recognized as the locus of long-term synaptic plasticity, which is correlated with learning and memory. The development of dendrites and spines in the mammalian central nervous system is a complex process that requires specific molecular events over a period of time. It has been shown that specific molecules are needed not only at the spine's point of contact, but also at a distance, providing signals that initiate a cascade of events leading to synapse formation. The specific molecules that act to signal neuronal differentiation, dendritic morphology, and synaptogenesis are tightly regulated by genetic and epigenetic programs. It has been shown that the dendritic spine structure and distribution are altered in many diseases, including many forms of mental retardation (MR), and can also be potentiated by neuronal activities and an enriched environment. Because dendritic spine pathologies are found in many types of MR, it has been proposed that an inability to form normal spines leads to the cognitive and motor deficits that are characteristic of MR. Epigenetic mechanisms, including DNA methylation, chromatin remodeling, and the noncoding RNA-mediated process, have profound regulatory roles in mammalian gene expression. The study of epigenetics focuses on cellular effects that result in a heritable pattern of gene expression without changes to genomic encoding. Despite extensive efforts to understand the molecular regulation of dendrite and spine development, epigenetic mechanisms have only recently been considered. In this review, we will focus on epigenetic mechanisms that regulate the development and maturation of dendrites and spines. We will discuss how epigenetic alterations could result in spine abnormalities that lead to MR, such as is seen in fragile X and Rett syndromes. We will also discuss both general methodology and recent technological advances in the study of neuronal dendrites and spines.
Keywords: dendritic spine; epigenetics; mental retardation; methyl-CpG binding protein 2 (MeCP2); microRNA; neurodevelopment; synapse.
Figures

Similar articles
-
Dendritic Spine and Synaptic Plasticity in Alzheimer's Disease: A Focus on MicroRNA.Front Cell Dev Biol. 2020 May 5;8:255. doi: 10.3389/fcell.2020.00255. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32432108 Free PMC article.
-
A genetic labeling system to study dendritic spine development in zebrafish models of neurodevelopmental disorders.Dis Model Mech. 2022 Aug 1;15(8):dmm049507. doi: 10.1242/dmm.049507. Epub 2022 Aug 19. Dis Model Mech. 2022. PMID: 35875841 Free PMC article.
-
βIII Spectrin Is Necessary for Formation of the Constricted Neck of Dendritic Spines and Regulation of Synaptic Activity in Neurons.J Neurosci. 2017 Jul 5;37(27):6442-6459. doi: 10.1523/JNEUROSCI.3520-16.2017. Epub 2017 Jun 2. J Neurosci. 2017. PMID: 28576936 Free PMC article.
-
Plasticity of dendritic spines: Molecular function and dysfunction in neurodevelopmental disorders.Psychiatry Clin Neurosci. 2019 Sep;73(9):541-550. doi: 10.1111/pcn.12899. Epub 2019 Jul 8. Psychiatry Clin Neurosci. 2019. PMID: 31215705 Review.
-
Dendritic spine pathology: cause or consequence of neurological disorders?Brain Res Brain Res Rev. 2002 Jun;39(1):29-54. doi: 10.1016/s0165-0173(02)00158-3. Brain Res Brain Res Rev. 2002. PMID: 12086707 Review.
Cited by
-
Gene Expressions Preferentially Influence Cortical Thickness of Human Connectome Project Atlas Parcellated Regions in First-Episode Antipsychotic-Naïve Psychoses.Schizophr Bull Open. 2023 Jul 19;4(1):sgad019. doi: 10.1093/schizbullopen/sgad019. eCollection 2023 Jan. Schizophr Bull Open. 2023. PMID: 37621304 Free PMC article.
-
Age-dependent expression of MeCP2 in a heterozygous mosaic mouse model.Hum Mol Genet. 2011 May 1;20(9):1834-43. doi: 10.1093/hmg/ddr066. Epub 2011 Feb 17. Hum Mol Genet. 2011. PMID: 21330301 Free PMC article.
-
DNA methylation as a putative mechanism for reduced dendritic spine density in the superior temporal gyrus of subjects with schizophrenia.Transl Psychiatry. 2017 Feb 14;7(2):e1032. doi: 10.1038/tp.2016.297. Transl Psychiatry. 2017. PMID: 28195572 Free PMC article.
-
Crosstalk among Epigenetic Pathways Regulates Neurogenesis.Front Neurosci. 2012 May 8;6:59. doi: 10.3389/fnins.2012.00059. eCollection 2012. Front Neurosci. 2012. PMID: 22586361 Free PMC article.
-
Fragile X Proteins FMRP and FXR2P Control Synaptic GluA1 Expression and Neuronal Maturation via Distinct Mechanisms.Cell Rep. 2015 Jun 16;11(10):1651-66. doi: 10.1016/j.celrep.2015.05.013. Epub 2015 Jun 4. Cell Rep. 2015. PMID: 26051932 Free PMC article.
References
-
- Akbarian S, Chen RZ, Gribnau J, Rasmussen TP, Fong H, Jaenisch R, Jones EG. Expression pattern of the Rett syndrome gene MeCP2 in primate prefrontal cortex. Neurobiol Dis. 2001;8(5):784–791. - PubMed
-
- Alvarez VA, Sabatini BL. Anatomical and physiological plasticity of dendritic spines. Annu Rev Neurosci. 2007;30:79–97. - PubMed
-
- Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet. 1999;23(2):185–188. - PubMed
-
- Antar LN, Dictenberg JB, Plociniak M, Afroz R, Bassell GJ. Localization of FMRP-associated mRNA granules and requirement of microtubules for activity-dependent trafficking in hippocampal neurons. Genes Brain Behav. 2005;4(6):350–359. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
