Cigarette smoke alters primary human bronchial epithelial cell differentiation at the air-liquid interface
- PMID: 25641363
- PMCID: PMC4313097
- DOI: 10.1038/srep08163
Cigarette smoke alters primary human bronchial epithelial cell differentiation at the air-liquid interface
Abstract
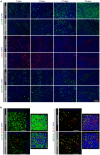
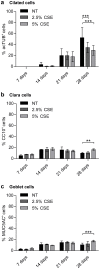
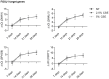
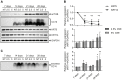
The differentiated human airway epithelium consists of different cell types forming a polarized and pseudostratified epithelium. This is dramatically altered in chronic obstructive pulmonary disease (COPD), characterized by basal and goblet cell hyperplasia, and squamous cell metaplasia. The effect of cigarette smoke on human bronchial epithelial cell (HBEC) differentiation remains to be elucidated. We analysed whether cigarette smoke extract (CSE) affected primary (p)HBEC differentiation and function. pHBEC were differentiated at the air-liquid interface (ALI) and differentiation was quantified after 7, 14, 21, or 28 days by assessing acetylated tubulin, CC10, or MUC5AC for ciliated, Clara, or goblet cells, respectively. Exposure of differentiating pHBEC to CSE impaired epithelial barrier formation, as assessed by resistance measurements (TEER). Importantly, CSE exposure significantly reduced the number of ciliated cells, while it increased the number of Clara and goblet cells. CSE-dependent cell number changes were reflected by a reduction of acetylated tubulin levels, an increased expression of the basal cell marker KRT14, and increased secretion of CC10, but not by changes in transcript levels of CC10, MUC5AC, or FOXJ1. Our data demonstrate that cigarette smoke specifically alters the cellular composition of the airway epithelium by affecting basal cell differentiation in a post-transcriptional manner.
Figures


References
-
- Staudt M. R. et al. Smoking Is Associated With A Loss Of Ciliated Cells Throughout The Airways. Am J Resp Crit Care Med - Meeting Abstracts 2014 189, A4097–A4097, 10.1164/ajrccm-conference.2014.189.1_MeetingAbstracts.A4097 (2014). - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

