Phosphoantigen Presentation to TCR γδ Cells, a Conundrum Getting Less Gray Zones
- PMID: 25642230
- PMCID: PMC4295553
- DOI: 10.3389/fimmu.2014.00679
Phosphoantigen Presentation to TCR γδ Cells, a Conundrum Getting Less Gray Zones
Abstract
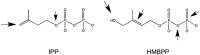
The mechanistic requirements of antigen recognition by T cells expressing a γδ TCR has revealed important differences with those of αβ TCR cells and, despite impressive new data generated in the very recent years, they remain poorly understood. Based on the structure of the TCR chains and the tissue distribution, γδ cells are represented in a variety of populations. The major subset of human peripheral blood γδ cells express Vγ9Vδ2 TCR heterodimers and are all stimulated by phosphorylated metabolites (commonly called phosphoantigens). Phosphoantigens are molecules with a very small mass and only stimulate Vγ9Vδ2 cells in the presence of antigen-presenting cells, suggesting a strict requirement for dedicated antigen-presenting molecules. Recent studies have identified butyrophilin (BTN) 3A1 as the molecule necessary to stimulate Vγ9Vδ2 cells. BTN3A1 extracellular, transmembrane, and cytoplasmic domains have different functions, including cognate interaction with the Vγ9Vδ2 TCR, binding of the phosphoantigens, and interaction with cytoplasmic proteins. This review mainly discusses the known molecular mechanisms of BTN3A1-mediated antigen presentation to γδ cells and proposes a model of phosphoantigen presentation, which integrates past and recent studies.
Keywords: antigen presentation; butyrophilin 3A1; infection control; tumor surveillance; γδ TCR.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

