Cofilin-induced unidirectional cooperative conformational changes in actin filaments revealed by high-speed atomic force microscopy
- PMID: 25642645
- PMCID: PMC4337605
- DOI: 10.7554/eLife.04806
Cofilin-induced unidirectional cooperative conformational changes in actin filaments revealed by high-speed atomic force microscopy
Abstract
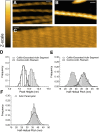
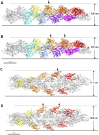
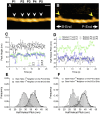
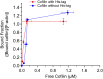
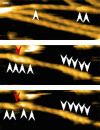
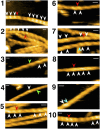
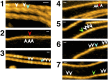
High-speed atomic force microscopy was employed to observe structural changes in actin filaments induced by cofilin binding. Consistent with previous electron and fluorescence microscopic studies, cofilin formed clusters along actin filaments, where the filaments were 2-nm thicker and the helical pitch was ~25% shorter, compared to control filaments. Interestingly, the shortened helical pitch was propagated to the neighboring bare zone on the pointed-end side of the cluster, while the pitch on the barbed-end side was similar to the control. Thus, cofilin clusters induce distinctively asymmetric conformational changes in filaments. Consistent with the idea that cofilin favors actin structures with a shorter helical pitch, cofilin clusters grew unidirectionally toward the pointed-end of the filament. Severing was often observed near the boundaries between bare zones and clusters, but not necessarily at the boundaries.
Keywords: S. cerevisiae; actin; biochemistry; biophysics; cofilin; cooperative conformational change; dictyostelium; human; myosin; structural biology.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures








Similar articles
-
Cofilin-induced cooperative conformational changes of actin subunits revealed using cofilin-actin fusion protein.Sci Rep. 2016 Feb 4;6:20406. doi: 10.1038/srep20406. Sci Rep. 2016. PMID: 26842224 Free PMC article.
-
Structures of cofilin-induced structural changes reveal local and asymmetric perturbations of actin filaments.Proc Natl Acad Sci U S A. 2020 Jan 21;117(3):1478-1484. doi: 10.1073/pnas.1915987117. Epub 2020 Jan 3. Proc Natl Acad Sci U S A. 2020. PMID: 31900364 Free PMC article.
-
Deciphering the actin structure-dependent preferential cooperative binding of cofilin.Elife. 2024 Aug 2;13:RP95257. doi: 10.7554/eLife.95257. Elife. 2024. PMID: 39093938 Free PMC article.
-
Biophysics of actin filament severing by cofilin.FEBS Lett. 2013 Apr 17;587(8):1215-9. doi: 10.1016/j.febslet.2013.01.062. Epub 2013 Feb 5. FEBS Lett. 2013. PMID: 23395798 Free PMC article. Review.
-
Actin dynamics and cofilin-actin rods in alzheimer disease.Cytoskeleton (Hoboken). 2016 Sep;73(9):477-97. doi: 10.1002/cm.21282. Epub 2016 Mar 1. Cytoskeleton (Hoboken). 2016. PMID: 26873625 Free PMC article. Review.
Cited by
-
Biophysics in Kanazawa University.Biophys Rev. 2020 Apr;12(2):249-251. doi: 10.1007/s12551-020-00635-2. Epub 2020 Feb 14. Biophys Rev. 2020. PMID: 32060734 Free PMC article. No abstract available.
-
Coronin Enhances Actin Filament Severing by Recruiting Cofilin to Filament Sides and Altering F-Actin Conformation.J Mol Biol. 2015 Sep 25;427(19):3137-47. doi: 10.1016/j.jmb.2015.08.011. Epub 2015 Aug 20. J Mol Biol. 2015. PMID: 26299936 Free PMC article.
-
Actin binding domain of filamin distinguishes posterior from anterior actin filaments in migrating Dictyostelium cells.Biophys Physicobiol. 2016 Dec 17;13:321-331. doi: 10.2142/biophysico.13.0_321. eCollection 2016. Biophys Physicobiol. 2016. PMID: 28409084 Free PMC article.
-
Purification and Ultramicroscopic Observation of the Influenza A Virus Ribonucleoprotein Complex.Methods Mol Biol. 2025;2890:141-149. doi: 10.1007/978-1-0716-4326-6_7. Methods Mol Biol. 2025. PMID: 39890725
-
Cofilactin filaments regulate filopodial structure and dynamics in neuronal growth cones.Nat Commun. 2022 May 4;13(1):2439. doi: 10.1038/s41467-022-30116-x. Nat Commun. 2022. PMID: 35508487 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

