Intestinal lipid-derived signals that sense dietary fat
- PMID: 25642767
- PMCID: PMC4362267
- DOI: 10.1172/JCI76302
Intestinal lipid-derived signals that sense dietary fat
Abstract
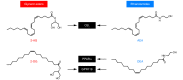
Fat is a vital macronutrient, and its intake is closely monitored by an array of molecular sensors distributed throughout the alimentary canal. In the mouth, dietary fat constituents such as mono- and diunsaturated fatty acids give rise to taste signals that stimulate food intake, in part by enhancing the production of lipid-derived endocannabinoid messengers in the gut. As fat-containing chyme enters the small intestine, it causes the formation of anorexic lipid mediators, such as oleoylethanolamide, which promote satiety. These anatomically and functionally distinct responses may contribute to the homeostatic control and, possibly, the pathological dysregulation of food intake.
Figures

Similar articles
-
Role of anorectic N-acylethanolamines in intestinal physiology and satiety control with respect to dietary fat.Pharmacol Res. 2014 Aug;86:18-25. doi: 10.1016/j.phrs.2014.03.006. Epub 2014 Mar 28. Pharmacol Res. 2014. PMID: 24681513 Review.
-
Bile acid composition regulates GPR119-dependent intestinal lipid sensing and food intake regulation in mice.Gut. 2020 Sep;69(9):1620-1628. doi: 10.1136/gutjnl-2019-319693. Epub 2020 Feb 28. Gut. 2020. PMID: 32111630 Free PMC article.
-
Fat, carbohydrate and protein by oral gavage in the rat can be equally effective for satiation.Physiol Behav. 2019 Aug 1;207:41-47. doi: 10.1016/j.physbeh.2019.04.022. Epub 2019 Apr 26. Physiol Behav. 2019. PMID: 31034840
-
The role of apolipoprotein A-IV in the regulation of food intake.Annu Rev Nutr. 2001;21:231-54. doi: 10.1146/annurev.nutr.21.1.231. Annu Rev Nutr. 2001. PMID: 11375436 Review.
-
High-fat hyperphagia in neurotrophin-4 deficient mice reveals potential role of vagal intestinal sensory innervation in long-term controls of food intake.Neurosci Lett. 2006 Jun 12;400(3):240-5. doi: 10.1016/j.neulet.2006.02.047. Epub 2006 Mar 13. Neurosci Lett. 2006. PMID: 16530962
Cited by
-
Functional Fiber Reduces Mice Obesity by Regulating Intestinal Microbiota.Nutrients. 2022 Jun 28;14(13):2676. doi: 10.3390/nu14132676. Nutrients. 2022. PMID: 35807856 Free PMC article.
-
Symphony of the gut microbiota and endocannabinoidome: a molecular and functional perspective.Front Cell Infect Microbiol. 2025 Mar 26;15:1566290. doi: 10.3389/fcimb.2025.1566290. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40207053 Free PMC article. Review.
-
Potential of dietary hemp and cannabinoids to modulate immune response to enhance health and performance in animals: opportunities and challenges.Front Immunol. 2023 Dec 4;14:1285052. doi: 10.3389/fimmu.2023.1285052. eCollection 2023. Front Immunol. 2023. PMID: 38111585 Free PMC article. Review.
-
Bile acids in glucose metabolism and insulin signalling - mechanisms and research needs.Nat Rev Endocrinol. 2019 Dec;15(12):701-712. doi: 10.1038/s41574-019-0266-7. Epub 2019 Oct 15. Nat Rev Endocrinol. 2019. PMID: 31616073 Free PMC article. Review.
-
Gut microbiota and oleoylethanolamide in the regulation of intestinal homeostasis.Front Endocrinol (Lausanne). 2023 Apr 5;14:1135157. doi: 10.3389/fendo.2023.1135157. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37091842 Free PMC article. Review.
References
-
- Leonard WR, Snodgrass JJ, Robertson ML. Evolutionary perspectives on fat ingestion and metabolism in humans. In: Montmayeur JP, le Coutre J, eds. Fat Detection: Taste, Texture, and Post Ingestive Effects. Boca Raton, Florida, USA: CRC Press; 2010:Chapter 1.
-
- Muskiet FAJ. Pathophysiology and evolutionary aspects of dietary fats and long-chain polyunsaturated fatty acids across the life cycle. In: Montmayeur JP, le Coutre J, eds. Fat Detection: Taste, Texture, and Post Ingestive Effects. Boca Raton, Florida, USA: CRC Press; 2010:Chapter 2. - PubMed

