Inactivation of DAP12 in PMN inhibits TREM1-mediated activation in rheumatoid arthritis
- PMID: 25642940
- PMCID: PMC4313943
- DOI: 10.1371/journal.pone.0115116
Inactivation of DAP12 in PMN inhibits TREM1-mediated activation in rheumatoid arthritis
Abstract
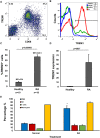
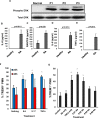
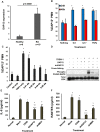
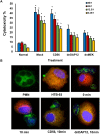
Rheumatoid arthritis (RA) is an autoimmune disease characterized by dysregulated and chronic systemic inflammatory responses that affect the synovium, bone, and cartilage causing damage to extra-articular tissue. Innate immunity is the first line of defense against invading pathogens and assists in the initiation of adaptive immune responses. Polymorphonuclear cells (PMNs), which include neutrophils, are the largest population of white blood cells in peripheral blood and functionally produce their inflammatory effect through phagocytosis, cytokine production and natural killer-like cytotoxic activity. TREM1 (triggering receptor expressed by myeloid cells) is an inflammatory receptor in PMNs that signals through the use of the intracellular activating adaptor DAP12 to induce downstream signaling. After TREM crosslinking, DAP12's tyrosines in its ITAM motif get phosphorylated inducing the recruitment of Syk tyrosine kinases and eventual activation of PI3 kinases and ERK signaling pathways. While both TREM1 and DAP12 have been shown to be important activators of RA pathogenesis, their activity in PMNs or the importance of DAP12 as a possible therapeutic target have not been shown. Here we corroborate, using primary RA specimens, that isolated PMNs have an increased proportion of both TREM1 and DAP12 compared to normal healthy control isolated PMNs both at the protein and gene expression levels. This increased expression is highly functional with increased activation of ERK and MAPKs, secretion of IL-8 and RANTES and cytotoxicity of target cells. Importantly, based on our hypothesis of an imbalance of activating and inhibitory signaling in the pathogenesis of RA we demonstrate that inhibition of the DAP12 signaling pathway inactivates these important inflammatory cells.
Conflict of interest statement
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

