Optogenetic perturbation of preBötzinger complex inhibitory neurons modulates respiratory pattern
- PMID: 25643296
- PMCID: PMC4340826
- DOI: 10.1038/nn.3938
Optogenetic perturbation of preBötzinger complex inhibitory neurons modulates respiratory pattern
Abstract
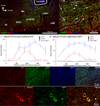
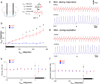
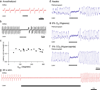
Inhibitory neurons make up a substantial fraction of the neurons in the preBötzinger complex (preBötC), a site that is critical for mammalian eupneic breathing. We investigated the role of glycinergic preBötC neurons in respiratory rhythmogenesis in mice using optogenetically targeted excitation and inhibition. Channelrhodopsin-2 (ChR2) or Archaerhodopsin (Arch) were expressed in glycinergic preBötC neurons of glycine transporter 2 (Glyt2, also known as Slc6a5)-Cre mice. In ChR2-transfected mice, brief inspiratory-phase bilateral photostimulation targeting the preBötC prematurely terminated inspiration, whereas expiratory-phase photostimulation delayed the onset of the next inspiration. Prolonged photostimulation produced apneas lasting as long as the light pulse. Inspiratory-phase photoinhibition in Arch-transfected mice during inspiration increased tidal volume without altering inspiratory duration, whereas expiratory-phase photoinhibition shortened the latency until the next inspiration. During persistent apneas, prolonged photoinhibition restored rhythmic breathing. We conclude that glycinergic preBötC neurons modulate inspiratory pattern and are important for reflex apneas, but that the rhythm can persist after substantial dampening of their activity.
Figures



References
-
- Stornetta RL, et al. A group of glutamatergic interneurons expressing high levels of both neurokinin-1 receptors and somatostatin identifies the region of the pre-Botzinger complex. J Comp Neurol. 2003;455:499–512. - PubMed
-
- Bouvier J, et al. Hindbrain interneurons and axon guidance signaling critical for breathing. Nat Neurosci. 2010;13:1066–1074. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

