ANGPTL3 is a novel biomarker as it activates ERK/MAPK pathway in oral cancer
- PMID: 25644496
- PMCID: PMC4430268
- DOI: 10.1002/cam4.418
ANGPTL3 is a novel biomarker as it activates ERK/MAPK pathway in oral cancer
Abstract
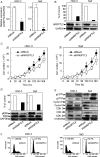
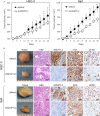
Angiopoietin-like 3 (ANGPTL3), which is involved in new blood vessel growth and stimulation of mitogen-activated protein kinase (MAPK), is expressed aberrantly in several types of human cancers. However, little is known about the relevance of ANGPTL3 in the behavior of oral squamous cell carcinoma (OSCC). In this study, we evaluated ANGPTL3 mRNA and protein in OSCC-derived cell lines (n = 8) and primary OSCCs (n = 109) and assessed the effect of ANGPTL3 on the biology and function of OSCCs in vitro and in vivo. Significant (P < 0.05) ANGPTL3 upregulation was detected in the cell lines and most primary OSCCs (60%) compared with the normal counterparts. The ANGPTL3 expression level was correlated closely (P < 0.05) with tumoral size. In patients with T3/T4 tumors, the overall survival rate with an ANGPTL3-positive tumor was significantly (P < 0.05) lower than that of ANGPTL3-negative cases. In vitro, cellular growth in ANGPTL3 knockdown cells significantly (P < 0.05) decreased with inactivated extracellular regulated kinase (ERK) and cell-cycle arrest at the G1 phase resulting from upregulation of the cyclin-dependent kinase inhibitors, including p21(Cip1) and p27(Kip1) . We also observed a marked (P < 0.05) reduction in the growth in ANGPTL3 knockdown-cell xenografts with decreased levels of phosphorylated ERK relative to control-cell xenografts. The current data indicated that ANGPTL3 may play a role in OSCCs via MAPK signaling cascades, making it a potentially useful diagnostic/therapeutic target for use in patients with OSCC.
Keywords: Angiopoietin-like 3; extracellular signal-regulated MAP Kinases; head and neck; squamous cell carcinoma; survival.
© 2015 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Figures


Similar articles
-
Persephin: A potential key component in human oral cancer progression through the RET receptor tyrosine kinase-mitogen-activated protein kinase signaling pathway.Mol Carcinog. 2015 Aug;54(8):608-17. doi: 10.1002/mc.22127. Epub 2013 Dec 23. Mol Carcinog. 2015. PMID: 24375483
-
Annexin A10 in human oral cancer: biomarker for tumoral growth via G1/S transition by targeting MAPK signaling pathways.PLoS One. 2012;7(9):e45510. doi: 10.1371/journal.pone.0045510. Epub 2012 Sep 17. PLoS One. 2012. PMID: 23029062 Free PMC article.
-
Cavin-2 in oral cancer: A potential predictor for tumor progression.Mol Carcinog. 2016 Jun;55(6):1037-47. doi: 10.1002/mc.22347. Epub 2015 Jun 18. Mol Carcinog. 2016. PMID: 26086332
-
ANGPTL3: a novel biomarker and promising therapeutic target.J Drug Target. 2019 Sep;27(8):876-884. doi: 10.1080/1061186X.2019.1566342. Epub 2019 Jan 25. J Drug Target. 2019. PMID: 30615486 Review.
-
MAPK Signaling Pathway in Oral Squamous Cell Carcinoma: Biological Function and Targeted Therapy.Cancers (Basel). 2022 Sep 23;14(19):4625. doi: 10.3390/cancers14194625. Cancers (Basel). 2022. PMID: 36230547 Free PMC article. Review.
Cited by
-
ANGPTL3 diminishes the resistance of ovarian cancer to paclitaxel by blocking the PI3K-AKT-mTOR signaling pathway.Heliyon. 2024 May 17;10(11):e31520. doi: 10.1016/j.heliyon.2024.e31520. eCollection 2024 Jun 15. Heliyon. 2024. PMID: 38828336 Free PMC article.
-
Angiopoietin-Like Proteins in Angiogenesis, Inflammation and Cancer.Int J Mol Sci. 2018 Feb 1;19(2):431. doi: 10.3390/ijms19020431. Int J Mol Sci. 2018. PMID: 29389861 Free PMC article. Review.
-
Angiopoietin-like protein 3: a novel potential biomarker for nephrotic syndrome in children.Front Pediatr. 2023 May 17;11:1113484. doi: 10.3389/fped.2023.1113484. eCollection 2023. Front Pediatr. 2023. PMID: 37266537 Free PMC article.
-
Evidence for Critical Role of Lymphocyte Cytosolic Protein 1 in Oral Cancer.Sci Rep. 2017 Feb 23;7:43379. doi: 10.1038/srep43379. Sci Rep. 2017. PMID: 28230172 Free PMC article.
-
Angiopoietin-like protein 3 blocks nuclear import of FAK and contributes to sorafenib response.Br J Cancer. 2018 Aug;119(4):450-461. doi: 10.1038/s41416-018-0189-4. Epub 2018 Jul 23. Br J Cancer. 2018. PMID: 30033448 Free PMC article.
References
-
- Camenisch G, Pisabarro MT, Sherman D, Kowalski J, Nagel M, Hass P, et al. ANGPTL3 stimulates endothelial cell adhesion and migration via integrin αvβ3 and induces blood vessel formation in vivo. J. Biol. Chem. 2002;277:17281–17290. - PubMed
-
- Li Y. Teng C. Angiopoietin-like proteins 3, 4 and 8: regulating lipid metabolism and providing new hope for metabolic syndrome. J. Drug Target. 2014;22:679–687. - PubMed
-
- Ono M, Shimizugawa T, Shimamura M, Yoshida K, Noji-Sakikawa C, Ando Y, et al. Protein region important for regulation of lipid metabolism in angiopoietin-like 3 (ANGPTL3) ANGPTL3 IS CLEAVED AND ACTIVATED IN VIVO. J. Biol. Chem. 2003;278:41804–41809. - PubMed
-
- Yu H, Zhang H, Li D, Xue H, Pan C, Zhao S, et al. Effects of ANGPTL3 antisense oligodeoxynucleotides transfection on the cell growths and invasion of human hepatocellular carcinoma cells. Hepatogastroenterology. 2011;58:1742–1746. - PubMed
-
- Chambard J-C, Lefloch R, Pouysségur J. Lenormand P. ERK implication in cell cycle regulation. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2007;1773:1299–1310. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

