Silencing of tuberin enhances photoreceptor survival and function in a preclinical model of retinitis pigmentosa (an american ophthalmological society thesis)
- PMID: 25646031
- PMCID: PMC4311672
Silencing of tuberin enhances photoreceptor survival and function in a preclinical model of retinitis pigmentosa (an american ophthalmological society thesis)
Abstract
Purpose: To assess the functional consequences of silencing of tuberin, an inhibitor of the mTOR signaling pathway, in a preclinical model of retinitis pigmentosa (RP) in order to test the hypothesis that insufficient induction of the protein kinase B (PKB)-regulated tuberin/mTOR self-survival pathway initiates apoptosis.
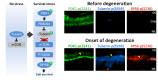
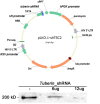
Methods: In an unbiased genome-scale approach, kinase peptide substrate arrays were used to analyze self-survival pathways at the onset of photoreceptor degeneration. The mutant Pde6b(H620Q)/Pde6b(H620Q) at P14 and P18 photoreceptor outer segment (OS) lysates were labeled with P-ATP and hybridized to an array of 1,164 different synthetic peptide substrates. At this stage, OS of Pde6b(H620Q)/Pde6b(H620Q) rods are morphologically normal. In vitro kinase assays and immunohistochemistry were used to validate phosphorylation. Short hairpin RNA (shRNA) gene silencing was used to validate tuberin's role in regulating survival.
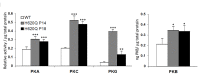
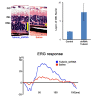
Results: At the onset of degeneration, 162 peptides were differentially phosphorylated. Protein kinases A, G, C (AGC kinases), and B exhibited increased activity in both peptide array and in vitro kinase assays. Immunohistochemical data confirmed altered phosphorylation patterns for phosphoinositide-dependent kinase-1 (PDK1), ribosomal protein S6 (RPS6), and tuberin. Tuberin gene silencing rescued photoreceptors from degeneration.
Conclusions: Phosphorylation of tuberin and RPS6 is due to the upregulated activity of PKB. PKB/tuberin cell growth/survival signaling is activated before the onset of degeneration. Substrates of the AGC kinases in the PKB/tuberin pathway are phosphorylated to promote cell survival. Knockdown of tuberin, the inhibitor of the mTOR pathway, increased photoreceptor survival and function in a preclinical model of RP.
Figures




Similar articles
-
Reprogramming towards anabolism impedes degeneration in a preclinical model of retinitis pigmentosa.Hum Mol Genet. 2016 Oct 1;25(19):4244-4255. doi: 10.1093/hmg/ddw256. Epub 2016 Aug 11. Hum Mol Genet. 2016. PMID: 27516389 Free PMC article.
-
Akt regulates nuclear/cytoplasmic localization of tuberin.Oncogene. 2007 Jan 25;26(4):521-31. doi: 10.1038/sj.onc.1209812. Epub 2006 Jul 24. Oncogene. 2007. PMID: 16862180
-
Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase.Proc Natl Acad Sci U S A. 2004 Sep 14;101(37):13489-94. doi: 10.1073/pnas.0405659101. Epub 2004 Sep 1. Proc Natl Acad Sci U S A. 2004. PMID: 15342917 Free PMC article.
-
Hamartin and tuberin: working together for tumour suppression.Int J Cancer. 2006 Jan 1;118(1):1-5. doi: 10.1002/ijc.21542. Int J Cancer. 2006. PMID: 16206276 Review.
-
Photoreceptor apoptosis in animal models. Implications for retinitis pigmentosa research.Arch Ophthalmol. 1995 Oct;113(10):1245-7. doi: 10.1001/archopht.1995.01100100033023. Arch Ophthalmol. 1995. PMID: 7575254 Review. No abstract available.
Cited by
-
Replenishment of TCA cycle intermediates provides photoreceptor resilience against neurodegeneration during progression of retinitis pigmentosa.JCI Insight. 2021 Sep 8;6(17):e150898. doi: 10.1172/jci.insight.150898. JCI Insight. 2021. PMID: 34292885 Free PMC article.
-
Peptide Arrays: Development and Application.Anal Chem. 2018 Jan 2;90(1):266-282. doi: 10.1021/acs.analchem.7b04380. Epub 2017 Dec 5. Anal Chem. 2018. PMID: 29135227 Free PMC article. Review. No abstract available.
-
Structural modeling of a novel SLC38A8 mutation that causes foveal hypoplasia.Mol Genet Genomic Med. 2017 Feb 26;5(3):202-209. doi: 10.1002/mgg3.266. eCollection 2017 May. Mol Genet Genomic Med. 2017. PMID: 28546991 Free PMC article.
-
Rapamycin Improved Retinal Function and Morphology in a Mouse Model of Retinal Degeneration.Front Neurosci. 2022 Feb 28;16:846584. doi: 10.3389/fnins.2022.846584. eCollection 2022. Front Neurosci. 2022. PMID: 35295093 Free PMC article.
-
Inhibition of mTOR signaling by rapamycin protects photoreceptors from degeneration in rd1 mice.Zool Res. 2021 Jul 18;42(4):482-486. doi: 10.24272/j.issn.2095-8137.2021.049. Zool Res. 2021. PMID: 34235896 Free PMC article.
References
-
- Berson EL. Retinitis pigmentosa: The Friedenwald lecture. Invest Ophthal Vis Sci. 1993;34(5):1659–1676. - PubMed
-
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368(9549):1795–1809. - PubMed
-
- Dinculescu A, Glushakova L, Min SH, Hauswirth WW. Adeno-associated virus-vectored gene therapy for retinal disease. Hum Gene Ther. 2005;16(6):649–663. - PubMed
-
- Schlichtenbrede FC, da Cruz L, Stephens C, et al. Long-term evaluation of retinal function in Prph2Rd2/Rd2 mice following AAV-mediated gene replacement therapy. J Gene Med. 2003;5(9):757–764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
