Identification and ultrastructural characterization of the Wolbachia symbiont in Litomosoides chagasfilhoi
- PMID: 25649218
- PMCID: PMC4323257
- DOI: 10.1186/s13071-015-0668-x
Identification and ultrastructural characterization of the Wolbachia symbiont in Litomosoides chagasfilhoi
Abstract
Background: Filarial nematodes are arthropod-transmitted parasites of vertebrates that affect more than 150 million people around the world and remain a major public health problem throughout tropical and subtropical regions. Despite the importance of these nematodes, the current treatment strategies are not efficient in eliminating the parasite. The main strategy of control is based on chemotherapy with diethylcarbamazine, albendazole and ivermectin. In the 1970s, it was found that some filarids possess endosymbiotic bacteria that are important for the development, survival and infectivity of the nematodes. These bacteria belong to the genus Wolbachia, which is a widespread and abundant intracellular symbiont in worms. Knowledge about the structure of the bacteria and their relationship with their nematode hosts may allow new perspectives for the control of filarial nematodes.
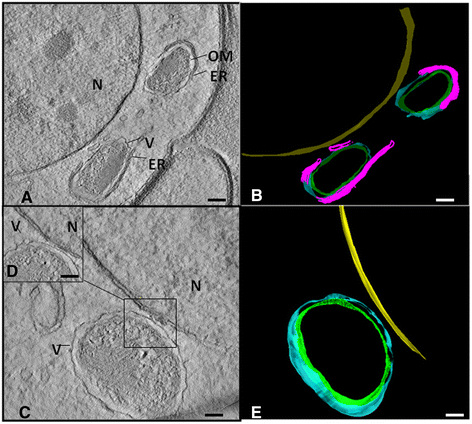
Methods: In this study, we used transmission electron microscopy combined with three-dimensional approaches to observe the structure of the endosymbiont of the filarial nematode Litomosoides chagasfilhoi, an experimental model for the study of lymphatic filariasis. In addition, the bacterium was classified based on PCR analyses.
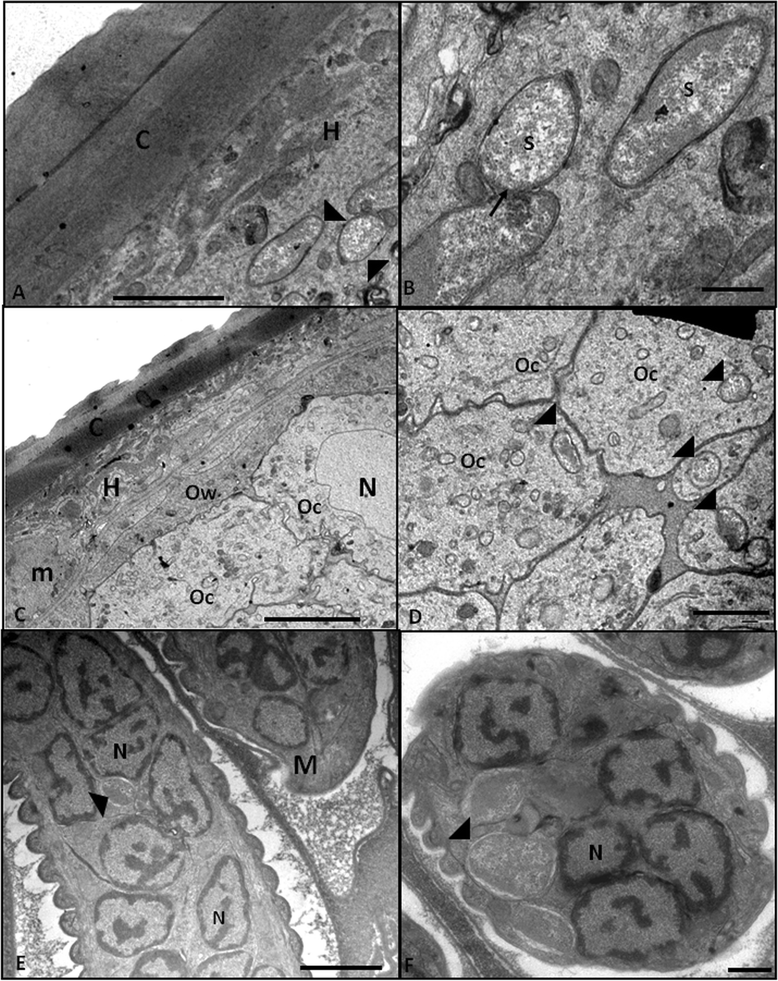
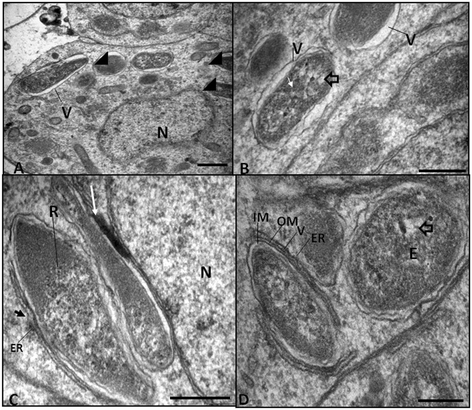
Results: The bacterium was mainly found in the hypodermis and in the female reproductive system in close association with host cell structures, such as the nucleus and endoplasmic reticulum. Our ultrastructural data also showed that the symbiont envelope is composed of two membrane units and is enclosed in a cytoplasmic vacuole, the symbiosome. Molecular data revealed that the bacterium of L. chagasfilhoi shares 100% identity with the Wolbachia endosymbiont of Litomosoides galizai.
Conclusions: Here we described ultrastructural aspects of the relationship of the Wolbachia with the filarial nematode Litomosoides chagasfilhoi and the findings lead us to consider this relationship as a mutualistic symbiosis.
Figures



Similar articles
-
Wolbachia filarial interactions.Cell Microbiol. 2013 Apr;15(4):520-6. doi: 10.1111/cmi.12084. Epub 2012 Dec 24. Cell Microbiol. 2013. PMID: 23210448 Review.
-
Wolbachia in filarial nematodes: evolutionary aspects and implications for the pathogenesis and treatment of filarial diseases.Vet Parasitol. 2001 Jul 12;98(1-3):215-38. doi: 10.1016/s0304-4017(01)00432-0. Vet Parasitol. 2001. PMID: 11516587 Review.
-
[Ultrastructural basis of interactions between prokaryotes and eukaryotes in different symbiotic models].Parassitologia. 2004 Jun;46(1-2):19-24. Parassitologia. 2004. PMID: 15305681 Review. Italian.
-
Trans-species transfer of Wolbachia: microinjection of Wolbachia from litomosoides sigmodontis into Acanthocheilonema viteae.Parasitology. 2003 Jun;126(Pt 6):503-11. Parasitology. 2003. PMID: 12866789
-
Wolbachia endosymbiotic bacteria of filarial nematodes. A new insight into disease pathogenesis and control.Arch Med Res. 2002 Jul-Aug;33(4):422-4. doi: 10.1016/s0188-4409(02)00377-6. Arch Med Res. 2002. PMID: 12234534
Cited by
-
Reliance of Wolbachia on High Rates of Host Proteolysis Revealed by a Genome-Wide RNAi Screen of Drosophila Cells.Genetics. 2017 Apr;205(4):1473-1488. doi: 10.1534/genetics.116.198903. Epub 2017 Feb 3. Genetics. 2017. PMID: 28159754 Free PMC article.
-
Differential gene expression in Drosophila melanogaster and D. nigrosparsa infected with the same Wolbachia strain.Sci Rep. 2021 May 31;11(1):11336. doi: 10.1038/s41598-021-90857-5. Sci Rep. 2021. PMID: 34059765 Free PMC article.
-
Wolbachia: endosymbiont of onchocercid nematodes and their vectors.Parasit Vectors. 2021 May 7;14(1):245. doi: 10.1186/s13071-021-04742-1. Parasit Vectors. 2021. PMID: 33962669 Free PMC article. Review.
-
Wbm0076, a candidate effector protein of the Wolbachia endosymbiont of Brugia malayi, disrupts eukaryotic actin dynamics.PLoS Pathog. 2023 Feb 17;19(2):e1010777. doi: 10.1371/journal.ppat.1010777. eCollection 2023 Feb. PLoS Pathog. 2023. PMID: 36800397 Free PMC article.
References
-
- Houerauf A, Volkmann L, Nissen-Paehle K, Schmetz C, Autenrieth I, Büttner DW, et al. Targeting Wolbachia endobacteria in Litomoroides sigmodontis: comparison of tetracyclines with chloramphenicol, macrolides and ciprofloxacin. Trop Med Int Health. 2000;5(4):275–9. doi: 10.1046/j.1365-3156.2000.00544.x. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

