Dynamic synchronization of ongoing neuronal activity across spinal segments regulates sensory information flow
- PMID: 25653206
- PMCID: PMC4457196
- DOI: 10.1113/jphysiol.2014.288134
Dynamic synchronization of ongoing neuronal activity across spinal segments regulates sensory information flow
Abstract
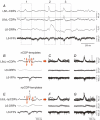
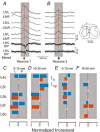
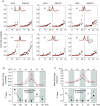
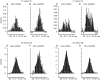
Previous studies on the correlation between spontaneous cord dorsum potentials recorded in the lumbar spinal segments of anaesthetized cats suggested the operation of a population of dorsal horn neurones that modulates, in a differential manner, transmission along pathways mediating Ib non-reciprocal postsynaptic inhibition and pathways mediating primary afferent depolarization and presynaptic inhibition. In order to gain further insight into the possible neuronal mechanisms that underlie this process, we have measured changes in the correlation between the spontaneous activity of individual dorsal horn neurones and the cord dorsum potentials associated with intermittent activation of these inhibitory pathways. We found that high levels of neuronal synchronization within the dorsal horn are associated with states of incremented activity along the pathways mediating presynaptic inhibition relative to pathways mediating Ib postsynaptic inhibition. It is suggested that ongoing changes in the patterns of functional connectivity within a distributed ensemble of dorsal horn neurones play a relevant role in the state-dependent modulation of impulse transmission along inhibitory pathways, among them those involved in the central control of sensory information. This feature would allow the same neuronal network to be involved in different functional tasks.
© 2015 The Authors. The Journal of Physiology © 2015 The Physiological Society.
Figures



Similar articles
-
Changes in correlation between spontaneous activity of dorsal horn neurones lead to differential recruitment of inhibitory pathways in the cat spinal cord.J Physiol. 2012 Apr 1;590(7):1563-84. doi: 10.1113/jphysiol.2011.223271. Epub 2012 Jan 23. J Physiol. 2012. PMID: 22271870 Free PMC article.
-
Supraspinal modulation of neuronal synchronization by nociceptive stimulation induces an enduring reorganization of dorsal horn neuronal connectivity.J Physiol. 2018 May 1;596(9):1747-1776. doi: 10.1113/JP275228. Epub 2018 Mar 26. J Physiol. 2018. PMID: 29451306 Free PMC article.
-
Modulation of synaptic transmission from segmental afferents by spontaneous activity of dorsal horn spinal neurones in the cat.J Physiol. 2000 Dec 1;529 Pt 2(Pt 2):445-60. doi: 10.1111/j.1469-7793.2000.00445.x. J Physiol. 2000. PMID: 11101653 Free PMC article.
-
Intersegmental synchronization of spontaneous activity of dorsal horn neurons in the cat spinal cord.Exp Brain Res. 2003 Feb;148(3):401-13. doi: 10.1007/s00221-002-1303-6. Epub 2002 Nov 28. Exp Brain Res. 2003. PMID: 12541150
-
Skin Innervation.J Invest Dermatol. 2024 Aug;144(8):1716-1723. doi: 10.1016/j.jid.2023.10.047. Epub 2024 Feb 24. J Invest Dermatol. 2024. PMID: 38402477 Review.
Cited by
-
Supraspinal Shaping of Adaptive Transitions in the State of Functional Connectivity Between Segmentally Distributed Dorsal Horn Neuronal Populations in Response to Nociception and Antinociception.Front Syst Neurosci. 2019 Sep 26;13:47. doi: 10.3389/fnsys.2019.00047. eCollection 2019. Front Syst Neurosci. 2019. PMID: 31616259 Free PMC article.
-
Analysis of spontaneous activity of superficial dorsal horn neurons in vitro: neuropathy-induced changes.Pflugers Arch. 2016 Nov;468(11-12):2017-2030. doi: 10.1007/s00424-016-1886-6. Epub 2016 Oct 10. Pflugers Arch. 2016. PMID: 27726011
-
Discrete field potentials produced by coherent activation of spinal dorsal horn neurons.Exp Brain Res. 2022 Feb;240(2):665-686. doi: 10.1007/s00221-021-06286-3. Epub 2022 Jan 10. Exp Brain Res. 2022. PMID: 35001174
-
A machine learning methodology for the selection and classification of spontaneous spinal cord dorsum potentials allows disclosure of structured (non-random) changes in neuronal connectivity induced by nociceptive stimulation.Front Neuroinform. 2015 Aug 26;9:21. doi: 10.3389/fninf.2015.00021. eCollection 2015. Front Neuroinform. 2015. PMID: 26379540 Free PMC article.
-
Markovian Analysis of the Sequential Behavior of the Spontaneous Spinal Cord Dorsum Potentials Induced by Acute Nociceptive Stimulation in the Anesthetized Cat.Front Comput Neurosci. 2017 May 1;11:32. doi: 10.3389/fncom.2017.00032. eCollection 2017. Front Comput Neurosci. 2017. PMID: 28507514 Free PMC article.
References
-
- Biella G, Riva L. Sotgiu ML. Interaction between neurons in different laminae of the dorsal horn of the spinal cord. A correlation study in normal and neuropathic rats. Eur J Neurosci. 1997;9:1017–1025. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

