The trichloroethylene metabolite S-(1,2-dichlorovinyl)-l-cysteine but not trichloroacetate inhibits pathogen-stimulated TNF-α in human extraplacental membranes in vitro
- PMID: 25653212
- PMCID: PMC4426062
- DOI: 10.1016/j.reprotox.2015.01.007
The trichloroethylene metabolite S-(1,2-dichlorovinyl)-l-cysteine but not trichloroacetate inhibits pathogen-stimulated TNF-α in human extraplacental membranes in vitro
Abstract
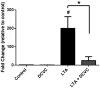
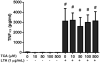
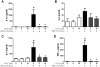
Extraplacental membranes define the gestational compartment and provide a barrier to infectious microorganisms ascending the gravid female reproductive tract. We tested the hypothesis that bioactive metabolites of trichloroethylene (TCE) decrease pathogen-stimulated innate immune response of extraplacental membranes. Extraplacental membranes were cultured for 4, 8, and 24h with the TCE metabolites trichloroacetate (TCA) or S-(1,2-dichlorovinyl)-l-cysteine (DCVC) in the absence or presence of lipoteichoic acid (LTA) or lipopolysaccharide (LPS) to simulate infection. In addition, membranes were cocultured with DCVC and Group B Streptococcus (GBS). DCVC (5-50μM) significantly inhibited LTA-, LPS-, and GBS-stimulated cytokine release from tissue cultures as early as 4h (P≤0.05). In contrast, TCA (up to 500μM) did not inhibit LTA-stimulated cytokine release from tissue punches. Because cytokines are important mediators for host response to infectious microorganisms these findings suggest that TCE exposure could potentially modify susceptibility to infection during pregnancy.
Keywords: Dichlorovinyl cysteine; Group B Streptococcus; Pregnancy; Toxicant–pathogen interactions; Trichloroethylene.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures
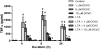
Similar articles
-
Role of cytokine signaling in group B Streptococcus-stimulated expression of human beta defensin-2 in human extraplacental membranes.Am J Reprod Immunol. 2015 Mar;73(3):263-72. doi: 10.1111/aji.12325. Epub 2014 Sep 29. Am J Reprod Immunol. 2015. PMID: 25263616 Free PMC article.
-
The antioxidant N-acetyl cysteine inhibits cytokine and prostaglandin release in human fetal membranes stimulated ex vivo with lipoteichoic acid or live group B streptococcus.Am J Reprod Immunol. 2024 Jan;91(1):e13807. doi: 10.1111/aji.13807. Am J Reprod Immunol. 2024. PMID: 38282602 Free PMC article.
-
Secretions of interleukin-1beta and tumor necrosis factor alpha by whole fetal membranes depend on initial interactions of amnion or choriodecidua with lipopolysaccharides or group B streptococci.Biol Reprod. 2004 Oct;71(4):1296-302. doi: 10.1095/biolreprod.104.028621. Epub 2004 Jun 16. Biol Reprod. 2004. PMID: 15201199
-
Mutagenicity of trichloroethylene and its metabolites: implications for the risk assessment of trichloroethylene.Environ Health Perspect. 2000 May;108 Suppl 2(Suppl 2):215-23. doi: 10.1289/ehp.00108s2215. Environ Health Perspect. 2000. PMID: 10807553 Free PMC article. Review.
-
Induction of pro-inflammatory cytokine gene expression and apoptosis in human chorion cells of fetal membranes by influenza virus infection: possible implications for maintenance and interruption of pregnancy during infection.Med Sci Monit. 2005 Jan;11(1):RA7-16. Med Sci Monit. 2005. PMID: 15614205 Review.
Cited by
-
Group B Streptococcal Maternal Colonization and Neonatal Disease: Molecular Mechanisms and Preventative Approaches.Front Pediatr. 2018 Feb 22;6:27. doi: 10.3389/fped.2018.00027. eCollection 2018. Front Pediatr. 2018. PMID: 29520354 Free PMC article. Review.
-
Trichloroethylene: An Update on an Environmental Contaminant with Multiple Health Effects.Annu Rev Pharmacol Toxicol. 2025 Jan;65(1):507-527. doi: 10.1146/annurev-pharmtox-022724-120525. Epub 2024 Dec 17. Annu Rev Pharmacol Toxicol. 2025. PMID: 39094062 Free PMC article. Review.
-
Placenta as a target of trichloroethylene toxicity.Environ Sci Process Impacts. 2020 Mar 1;22(3):472-486. doi: 10.1039/c9em00537d. Epub 2020 Feb 5. Environ Sci Process Impacts. 2020. PMID: 32022077 Free PMC article.
-
Evaluating impacts of the trichloroethylene metabolite S-(1,2-dichlorovyinyl)-L-cysteine on transcriptomic responses and cytokine release in a macrophage model: implications for pregnancy outcomes.J Immunotoxicol. 2025 Dec;22(1):2522041. doi: 10.1080/1547691X.2025.2522041. Epub 2025 Jul 3. J Immunotoxicol. 2025. PMID: 40607769
-
Trichloroethylene exposure in mid-pregnancy decreased fetal weight and increased placental markers of oxidative stress in rats.Reprod Toxicol. 2019 Jan;83:38-45. doi: 10.1016/j.reprotox.2018.11.002. Epub 2018 Nov 20. Reprod Toxicol. 2019. PMID: 30468822 Free PMC article.
References
-
- Goldenberg RL, Hauth JC, Andrews WW. Intrauterine infection and preterm delivery. N Engl J Med. 2000;342(20):1500–7. - PubMed
-
- Romero R, Mazor M, Tartakovsky B. Systemic administration of interleukin-1 induces preterm parturition in mice. Am J Obstet Gynecol. 1991;165(4 Pt 1):969–71. - PubMed
-
- Pararas MV, Skevaki CL, Kafetzis DA. Preterm birth due to maternal infection: Causative pathogens and modes of prevention. Eur J Clin Microbiol Infect Dis. 2006;25(9):562–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

