Mitochondrial protection by the mixed muscarinic/σ1 ligand ANAVEX2-73, a tetrahydrofuran derivative, in Aβ25-35 peptide-injected mice, a nontransgenic Alzheimer's disease model
- PMID: 25653589
- PMCID: PMC4299448
- DOI: 10.3389/fncel.2014.00463
Mitochondrial protection by the mixed muscarinic/σ1 ligand ANAVEX2-73, a tetrahydrofuran derivative, in Aβ25-35 peptide-injected mice, a nontransgenic Alzheimer's disease model
Abstract
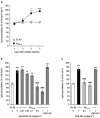
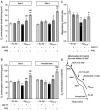
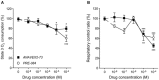
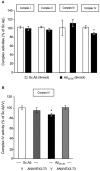
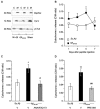
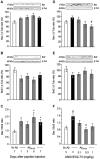
Alzheimer's disease (AD), the most prevalent dementia in the elderly, is characterized by progressive synaptic and neuronal loss. Mitochondrial dysfunctions have been consistently reported as an early event in AD and appear before Aβ deposition and memory decline. In order to define a new neuroprotectant strategy in AD targeting mitochondrial alterations, we develop tetrahydro-N,N-dimethyl-2,2-diphenyl-3-furanmethanamine (ANAVEX2-73, AE37), a mixed muscarinic receptor ligand and a sigma-1 receptor (σ1R) agonist. We previously reported that ANAVEX2-73 shows anti-amnesic and neuroprotective activities in mice injected intracerebroventricular (ICV) with oligomeric amyloid-β25-35 peptide (Aβ25-35). The σ1R is present at mitochondria-associated endoplasmic reticulum (ER) membranes, where it acts as a sensor/modulator of ER stress responses and local Ca(2+) exchanges with the mitochondria. We therefore evaluated the effect of ANAVEX2-73 and PRE-084, a reference σ1R agonist, on preservation of mitochondrial integrity in Aβ25-35-injected mice. In isolated mitochondria from hippocampus preparations of Aβ25-35 injected animals, we measured respiration rates, complex activities, lipid peroxidation, Bax/Bcl-2 ratios and cytochrome c release into the cytosol. Five days after Aβ25-35 injection, mitochondrial respiration in mouse hippocampus was altered. ANAVEX2-73 (0.01-1 mg/kg IP) restored normal respiration and PRE-084 (0.5-1 mg/kg IP) increased respiration rates. Both compounds prevented Aβ25-35-induced increases in lipid peroxidation levels, Bax/Bcl-2 ratio and cytochrome c release into the cytosol, all indicators of increased toxicity. ANAVEX2-73 and PRE-084 efficiently prevented the mitochondrial respiratory dysfunction and resulting oxidative stress and apoptosis. The σ1R, targeted selectively or non-selectively, therefore appears as a valuable target for protection against mitochondrial damages in AD.
Keywords: ANAVEX2-73; Alzheimer’s disease; cytoprotection; mitochondrial damages; sigma-1 receptor.
Figures
References
-
- Aleardi A. M., Benard G., Augereau O., Malgat M., Talbot J. C., Mazat J. P., et al. (2005). Gradual alteration of mitochondrial structure and function by beta-amyloids: importance of membrane viscosity changes, energy deprivation, reactive oxygen species production and cytochrome c release. J. Bioenerg. Biomembr. 37, 207–225. 10.1007/s10863-005-6631-3 - DOI - PubMed
-
- Bobba A., Amadoro G., Valenti D., Corsetti V., Lassandro R., Atlante A. (2013). Mitochondrial respiratory chain Complexes I and IV are impaired by β-amyloid via direct interaction and through Complex I-dependent ROS production, respectively. Mitochondrion 13, 298–311. 10.1016/j.mito.2013.03.008 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

