Porphyromonas gingivalis virulence factors involved in subversion of leukocytes and microbial dysbiosis
- PMID: 25654623
- PMCID: PMC4601496
- DOI: 10.1080/21505594.2014.999567
Porphyromonas gingivalis virulence factors involved in subversion of leukocytes and microbial dysbiosis
Abstract
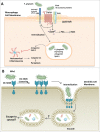
The oral bacterium Porphyromonas gingivalis has special nutrient requirements due to its asaccharolytic nature subsisting on small peptides cleaved from host proteins. Using proteases and other virulence factors, P. gingivalis thrives as a component of a polymicrobial community in nutritionally favorable inflammatory environments. In this regard, P. gingivalis has a number of strategies that subvert the host immune response in ways that promote its colonization and facilitate the outgrowth of the surrounding microbial community. The focus of this review is to discuss at the molecular level how P. gingivalis subverts leukocytes to create a favorable environment for a select community of bacteria that, in turn, adversely affects the periodontal tissues.
Keywords: P. gingivalis; dysbiosis; immune subversion; inflammation leukocytes; periodontitis.
Figures
Similar articles
-
Roles of Porphyromonas gingivalis and its virulence factors in periodontitis.Adv Protein Chem Struct Biol. 2020;120:45-84. doi: 10.1016/bs.apcsb.2019.12.001. Epub 2020 Jan 10. Adv Protein Chem Struct Biol. 2020. PMID: 32085888 Free PMC article. Review.
-
Porphyromonas gingivalis: an invasive and evasive opportunistic oral pathogen.FEMS Microbiol Lett. 2012 Aug;333(1):1-9. doi: 10.1111/j.1574-6968.2012.02579.x. Epub 2012 May 28. FEMS Microbiol Lett. 2012. PMID: 22530835 Review.
-
Regulation of tryptophan-indole metabolic pathway in Porphyromonas gingivalis virulence and microbiota dysbiosis in periodontitis.NPJ Biofilms Microbiomes. 2025 Feb 27;11(1):37. doi: 10.1038/s41522-025-00669-y. NPJ Biofilms Microbiomes. 2025. PMID: 40011497 Free PMC article.
-
Genetic diversity in the oral pathogen Porphyromonas gingivalis: molecular mechanisms and biological consequences.Future Microbiol. 2013 May;8(5):607-20. doi: 10.2217/fmb.13.30. Future Microbiol. 2013. PMID: 23642116 Free PMC article. Review.
-
Molecular Strategies Underlying Porphyromonas gingivalis Virulence.J Mol Biol. 2021 Apr 2;433(7):166836. doi: 10.1016/j.jmb.2021.166836. Epub 2021 Feb 1. J Mol Biol. 2021. PMID: 33539891 Review.
Cited by
-
DAMPs and alarmin gene expression patterns in aging healthy and diseased mucosal tissues.Front Oral Health. 2023 Nov 30;4:1320083. doi: 10.3389/froh.2023.1320083. eCollection 2023. Front Oral Health. 2023. PMID: 38098978 Free PMC article.
-
The Distinct Immune-Stimulatory Capacities of Porphyromonas gingivalis Strains 381 and ATCC 33277 Are Determined by the fimB Allele and Gingipain Activity.Infect Immun. 2019 Nov 18;87(12):e00319-19. doi: 10.1128/IAI.00319-19. Print 2019 Dec. Infect Immun. 2019. PMID: 31570556 Free PMC article.
-
Effects of estradiol on the virulence traits of Porphyromonas gingivalis.Sci Rep. 2022 Aug 16;12(1):13881. doi: 10.1038/s41598-022-17019-z. Sci Rep. 2022. PMID: 35974048 Free PMC article.
-
Microbiological analysis and the outcomes of periodontal treatment with or without adjunctive systemic antibiotics-a retrospective study.Clin Oral Investig. 2018 Dec;22(9):3031-3041. doi: 10.1007/s00784-018-2392-3. Epub 2018 Feb 21. Clin Oral Investig. 2018. PMID: 29468598
-
Oral microbiota in the oral-genitourinary axis: identifying periodontitis as a potential risk of genitourinary cancers.Mil Med Res. 2021 Sep 29;8(1):54. doi: 10.1186/s40779-021-00344-1. Mil Med Res. 2021. PMID: 34588004 Free PMC article. Review.
References
-
- Pihlstrom BL, Michalowicz BS, Johnson NW. Periodontal diseases. Lancet 2005; 366:1809-20; PMID:16298220; http://dx.doi.org/10.1016/S0140-6736(05)67728-8 - DOI - PubMed
-
- Hajishengallis G. Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol 2015; 15(1):30-44; PMID:25534621; http://dx.doi.org/10.1038/nri3785 - DOI - PMC - PubMed
-
- Abusleme L, Dupuy AK, Dutzan N, Silva N, Burleson JA, Strausbaugh LD, Gamonal J, Diaz PI. The subgingival microbiome in health and periodontitis and its relationship with community biomass and inflammation. ISME J 2013; 7:1016-25; PMID:23303375; http://dx.doi.org/10.1038/ismej.2012.174 - DOI - PMC - PubMed
-
- Jorth P, Turner KH, Gumus P, Nizam N, Buduneli N, Whiteley M. Metatranscriptomics of the human oral microbiome during health and disease. MBio 2014; 5:e01012-14; PMID:24692635; http://dx.doi.org/10.1128/mBio.01012-14 - DOI - PMC - PubMed
-
- Hajishengallis G, Lamont RJ. Beyond the red complex and into more complexity: The Polymicrobial Synergy and Dysbiosis (PSD) model of periodontal disease etiology. Mol Oral Microbiol 2012; 27:409-19; PMID:23134607; http://dx.doi.org/10.1111/j.2041-1014.2012.00663.x - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
