Non-contact acoustic radiation force impulse microscopy via photoacoustic detection for probing breast cancer cell mechanics
- PMID: 25657870
- PMCID: PMC4317122
- DOI: 10.1364/BOE.6.000011
Non-contact acoustic radiation force impulse microscopy via photoacoustic detection for probing breast cancer cell mechanics
Abstract
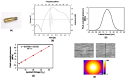
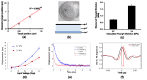
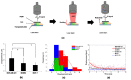
We demonstrate a novel non-contact method: acoustic radiation force impulse microscopy via photoacoustic detection (PA-ARFI), capable of probing cell mechanics. A 30 MHz lithium niobate ultrasound transducer is utilized for both detection of phatoacoustic signals and generation of acoustic radiation force. To track cell membrane displacements by acoustic radiation force, functionalized single-walled carbon nanotubes are attached to cell membrane. Using the developed microscopy evaluated with agar phantoms, the mechanics of highly- and weakly-metastatic breast cancer cells are quantified. These results clearly show that the PA-ARFI microscopy may serve as a novel tool to probe mechanics of single breast cancer cells.
Keywords: (170.0180) Microscopy; (170.5120) Photoacoustic imaging; (170.7170) Ultrasound.
Figures

Similar articles
-
Jitter reduction technique for acoustic radiation force impulse microscopy via photoacoustic detection.Opt Express. 2015 Jul 27;23(15):19166-75. doi: 10.1364/OE.23.019166. Opt Express. 2015. PMID: 26367579 Free PMC article.
-
Evaluating the intensity of the acoustic radiation force impulse (ARFI) in intravascular ultrasound (IVUS) imaging: Preliminary in vitro results.Ultrasonics. 2016 Aug;70:64-74. doi: 10.1016/j.ultras.2016.04.017. Epub 2016 Apr 20. Ultrasonics. 2016. PMID: 27135187
-
Acoustic radiation force impulse (ARFI) imaging of zebrafish embryo by high-frequency coded excitation sequence.Ann Biomed Eng. 2012 Apr;40(4):907-15. doi: 10.1007/s10439-011-0466-3. Epub 2011 Nov 19. Ann Biomed Eng. 2012. PMID: 22101757 Free PMC article.
-
Ultrasound Acoustic Radiation Force Impulse imaging for High Intensity Focused Ultrasound focal spot localization.Annu Int Conf IEEE Eng Med Biol Soc. 2018 Jul;2018:5713-5716. doi: 10.1109/EMBC.2018.8513591. Annu Int Conf IEEE Eng Med Biol Soc. 2018. PMID: 30441633
-
Acoustic radiation force impulse elastography for differentiation of malignant and benign breast lesions: a meta-analysis.Int J Clin Exp Med. 2015 Apr 15;8(4):4753-61. eCollection 2015. Int J Clin Exp Med. 2015. PMID: 26131049 Free PMC article. Review.
Cited by
-
Cell Deformation by Single-beam Acoustic Trapping: A Promising Tool for Measurements of Cell Mechanics.Sci Rep. 2016 Jun 8;6:27238. doi: 10.1038/srep27238. Sci Rep. 2016. PMID: 27273365 Free PMC article.
-
Multi-parametric Photoacoustic Imaging Combined with Acoustic Radiation Force Impulse Imaging for Applications in Tissue Engineering.Ann Biomed Eng. 2025 Feb;53(2):371-382. doi: 10.1007/s10439-024-03617-7. Epub 2024 Sep 18. Ann Biomed Eng. 2025. PMID: 39294465
-
Jitter reduction technique for acoustic radiation force impulse microscopy via photoacoustic detection.Opt Express. 2015 Jul 27;23(15):19166-75. doi: 10.1364/OE.23.019166. Opt Express. 2015. PMID: 26367579 Free PMC article.
-
A Novel Fisheye-Lens-Based Photoacoustic System.Sensors (Basel). 2016 Dec 19;16(12):2185. doi: 10.3390/s16122185. Sensors (Basel). 2016. PMID: 27999376 Free PMC article.
-
Label-free analysis of the characteristics of a single cell trapped by acoustic tweezers.Sci Rep. 2017 Oct 26;7(1):14092. doi: 10.1038/s41598-017-14572-w. Sci Rep. 2017. PMID: 29074938 Free PMC article.
References
-
- Wang Y. L., Discher D. E., Cell Mechanics (Academic Press, San Diego, 2007).
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
