Long-lasting effects of early-life antibiotic treatment and routine animal handling on gut microbiota composition and immune system in pigs
- PMID: 25658611
- PMCID: PMC4319779
- DOI: 10.1371/journal.pone.0116523
Long-lasting effects of early-life antibiotic treatment and routine animal handling on gut microbiota composition and immune system in pigs
Abstract
Background: In intensive pig husbandry systems, antibiotics are frequently administrated during early life stages to prevent respiratory and gastro-intestinal tract infections, often in combination with stressful handlings. The immediate effects of these treatments on microbial colonization and immune development have been described recently. Here we studied whether the early life administration of antibiotics has long-lasting effects on the pig's intestinal microbial community and on gut functionality.
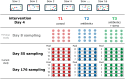
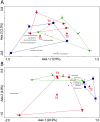
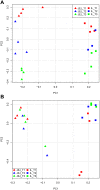
Methodology/principal findings: To investigate the long-lasting effect of early-life treatment, piglets were divided into three different groups receiving the following treatments: 1) no antibiotics and no stress, 2) antibiotics and no stress, and 3) antibiotics and stress. All treatments were applied at day four after birth. Sampling of jejunal content for community scale microbiota analysis, and jejunal and ileal tissue for genome-wide transcription profiling, was performed at day 55 (~8 weeks) and day 176 (~25 weeks) after birth. Antibiotic treatment in combination with or without exposure to stress was found to have long-lasting effects on host intestinal gene expression involved in a multitude of processes, including immune related processes.
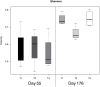
Conclusions/significance: The results obtained in this study indicate that early life (day 4 after birth) perturbations have long-lasting effects on the gut system, both in gene expression (day 55) as well as on microbiota composition (day 176). At day 55 high variance was observed in the microbiota data, but no significant differences between treatment groups, which is most probably due to the newly acquired microbiota during and right after weaning (day 28). Based on the observed difference in gene expression at day 55, it is hypothesized that due to the difference in immune programming during early life, the systems respond differently to the post-weaning newly acquired microbiota. As a consequence, the gut systems of the treatment groups develop into different homeostasis.
Conflict of interest statement
Figures

Similar articles
-
Early-life environmental variation affects intestinal microbiota and immune development in new-born piglets.PLoS One. 2014 Jun 18;9(6):e100040. doi: 10.1371/journal.pone.0100040. eCollection 2014. PLoS One. 2014. PMID: 24941112 Free PMC article.
-
Perturbation of microbiota in one-day old broiler chickens with antibiotic for 24 hours negatively affects intestinal immune development.BMC Genomics. 2017 Mar 20;18(1):241. doi: 10.1186/s12864-017-3625-6. BMC Genomics. 2017. PMID: 28320307 Free PMC article.
-
Dietary grape seed proanthocyanidins (GSPs) improve weaned intestinal microbiota and mucosal barrier using a piglet model.Oncotarget. 2016 Dec 6;7(49):80313-80326. doi: 10.18632/oncotarget.13450. Oncotarget. 2016. PMID: 27880936 Free PMC article.
-
Antibiotics and the developing infant gut microbiota and resistome.Curr Opin Microbiol. 2015 Oct;27:51-6. doi: 10.1016/j.mib.2015.07.007. Epub 2015 Aug 1. Curr Opin Microbiol. 2015. PMID: 26241507 Free PMC article. Review.
-
Antibiotic effects on gut microbiota, metabolism, and beyond.Appl Microbiol Biotechnol. 2019 Dec;103(23-24):9277-9285. doi: 10.1007/s00253-019-10165-x. Epub 2019 Nov 7. Appl Microbiol Biotechnol. 2019. PMID: 31701196 Review.
Cited by
-
Maternal antibiotic treatment affects offspring gastric sensing for umami taste and ghrelin regulation in the pig.J Anim Sci Biotechnol. 2021 Mar 18;12(1):31. doi: 10.1186/s40104-021-00557-3. J Anim Sci Biotechnol. 2021. PMID: 33731211 Free PMC article.
-
High-level integration of murine intestinal transcriptomics data highlights the importance of the complement system in mucosal homeostasis.BMC Genomics. 2019 Dec 30;20(1):1028. doi: 10.1186/s12864-019-6390-x. BMC Genomics. 2019. PMID: 31888466 Free PMC article.
-
Fecal Microbiota and Its Correlation With Fatty Acids and Free Amino Acids Metabolism in Piglets After a Lactobacillus Strain Oral Administration.Front Microbiol. 2019 Apr 16;10:785. doi: 10.3389/fmicb.2019.00785. eCollection 2019. Front Microbiol. 2019. PMID: 31040835 Free PMC article.
-
The effect of maternal antibiotic use in sows on intestinal development in offspring.J Anim Sci. 2020 Jun 1;98(6):skaa181. doi: 10.1093/jas/skaa181. J Anim Sci. 2020. PMID: 32479635 Free PMC article.
-
Neonatal and maternal dietary interventions driving microbiota and functionality in piglet gut compartments.Sci Rep. 2025 Feb 25;15(1):6771. doi: 10.1038/s41598-025-90781-y. Sci Rep. 2025. PMID: 40000703 Free PMC article.
References
-
- Starke IC, Pieper R, Neumann K, Zentek J, Vahjen W (2013) The impact of high dietary zinc oxide on the development of the intestinal microbiota in weaned piglets. FEMS Microbiol Ecol. - PubMed
-
- Yao K, Sun Z, Liu Z, Li Z, Yin Y (2013) Development of the Gastrointestinal Tract in Pigs. Nutritional and Physiological Functions of Amino Acids in Pigs: 3–18.
-
- Geuking MB, McCoy KD, Macpherson AJ (2011) The continuum of intestinal CD4+ T cell adaptations in host-microbial mutualism. Gut Microbes 2. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

