Induction of PLSCR1 in a STING/IRF3-dependent manner upon vector transfection in ovarian epithelial cells
- PMID: 25658875
- PMCID: PMC4320088
- DOI: 10.1371/journal.pone.0117464
Induction of PLSCR1 in a STING/IRF3-dependent manner upon vector transfection in ovarian epithelial cells
Abstract
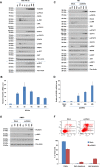
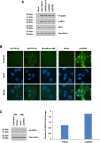
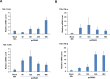
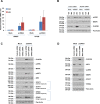
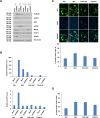
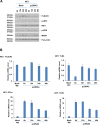
Toll-like receptors (TLRs) are the primary sensors of the innate immune system that recognize pathogenic nucleic acids including double-stranded plasmid DNA (dsDNA). TLR signaling activates multiple pathways including IRF3 which is involved in transcriptional induction of inflammatory cytokines (i.e. interferons (IFNs)). Phospholipid scramblase 1, PLSCR1, is a highly inducible IFN-regulated gene mediating anti-viral properties of IFNs. Herein, we report a novel finding that dsDNA transfection in T80 immortalized normal ovarian surface epithelial cell line leads to a marked increase in PLSCR1 mRNA and protein. We also noted a comparable response in primary mammary epithelial cells (HMECs). Similar to IFN-2α treated cells, de novo synthesized PLSCR1 was localized predominantly to the plasma membrane. dsDNA transfection, in T80 and HMEC cells, led to activation of MAPK and IRF3. Although inhibition of MAPK (using U0126) did not modulate PLSCR1 mRNA and protein, IRF3 knockdown (using siRNA) significantly ablated the PLSCR1 induction. In prior studies, the activation of IRF3 was shown to be mediated by cGAS-STING pathway. To investigate the contribution of STING to PLSCR1 induction, we utilized siRNA to reduce STING expression and observed that PLSCR1 protein was markedly reduced. In contrast to normal T80/HMECs, the phosphorylation of IRF3 as well as induction of STING and PLSCR1 were absent in ovarian cancer cells (serous, clear cell, and endometrioid) suggesting that the STING/IRF3 pathway may be dysregulated in these cancer cells. However, we also noted induction of different TLR and IFN mRNAs between the T80 and HEY (serous epithelial ovarian carcinoma) cell lines upon dsDNA transfection. Collectively, these results indicate that the STING/IRF3 pathway, activated following dsDNA transfection, contributes to upregulation of PLSCR1 in ovarian epithelial cells.
Conflict of interest statement
Figures

Similar articles
-
Interferon-alpha-induced expression of phospholipid scramblase 1 through STAT1 requires the sequential activation of protein kinase Cdelta and JNK.J Biol Chem. 2005 Dec 30;280(52):42707-14. doi: 10.1074/jbc.M506178200. Epub 2005 Oct 28. J Biol Chem. 2005. PMID: 16260419
-
Activation of the cGAS-STING-IRF3 Axis by Type I and II Interferons Contributes to Host Defense.Adv Sci (Weinh). 2024 Sep;11(35):e2308890. doi: 10.1002/advs.202308890. Epub 2024 Jul 14. Adv Sci (Weinh). 2024. PMID: 39004913 Free PMC article.
-
Downstream STING pathways IRF3 and NF-κB differentially regulate CCL22 in response to cytosolic dsDNA.Cancer Gene Ther. 2024 Jan;31(1):28-42. doi: 10.1038/s41417-023-00678-z. Epub 2023 Nov 21. Cancer Gene Ther. 2024. PMID: 37990062
-
Phospholipid scramblase 1: a frontline defense against viral infections.Front Cell Infect Microbiol. 2025 Apr 3;15:1573373. doi: 10.3389/fcimb.2025.1573373. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40248364 Free PMC article. Review.
-
Phospholipid scramblase 1: a protein with multiple functions via multiple molecular interactors.Cell Commun Signal. 2022 Jun 1;20(1):78. doi: 10.1186/s12964-022-00895-3. Cell Commun Signal. 2022. PMID: 35650588 Free PMC article. Review.
Cited by
-
The Interferon-Inducible Human PLSCR1 Protein Is a Restriction Factor of Human Cytomegalovirus.Microbiol Spectr. 2022 Feb 23;10(1):e0134221. doi: 10.1128/spectrum.01342-21. Epub 2022 Feb 9. Microbiol Spectr. 2022. PMID: 35138119 Free PMC article.
-
STING Signaling in Cancer Cells: Important or Not?Arch Immunol Ther Exp (Warsz). 2018 Apr;66(2):125-132. doi: 10.1007/s00005-017-0481-7. Epub 2017 Jul 26. Arch Immunol Ther Exp (Warsz). 2018. PMID: 28748479 Free PMC article. Review.
-
zDHHC-Mediated S-Palmitoylation in Skin Health and Its Targeting as a Treatment Perspective.Int J Mol Sci. 2025 Feb 15;26(4):1673. doi: 10.3390/ijms26041673. Int J Mol Sci. 2025. PMID: 40004137 Free PMC article. Review.
-
Regulating Immune Responses Induced by PEGylated Messenger RNA-Lipid Nanoparticle Vaccine.Vaccines (Basel). 2024 Dec 27;13(1):14. doi: 10.3390/vaccines13010014. Vaccines (Basel). 2024. PMID: 39852793 Free PMC article. Review.
-
Phospholipid Scramblases: Role in Cancer Progression and Anticancer Therapeutics.Front Genet. 2022 Mar 29;13:875894. doi: 10.3389/fgene.2022.875894. eCollection 2022. Front Genet. 2022. PMID: 35422844 Free PMC article. Review.
References
-
- Midoux P, Breuzard G, Gomez JP, Pichon C (2008) Polymer-based gene delivery: a current review on the uptake and intracellular trafficking of polyplexes. Curr Gene Ther 8: 335–352. - PubMed
-
- Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, et al. (2004) Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 303: 1526–1529. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

