Feasibility and utility of applications of the common data model to multiple, disparate observational health databases
- PMID: 25670757
- PMCID: PMC4457111
- DOI: 10.1093/jamia/ocu023
Feasibility and utility of applications of the common data model to multiple, disparate observational health databases
Abstract
Objectives: To evaluate the utility of applying the Observational Medical Outcomes Partnership (OMOP) Common Data Model (CDM) across multiple observational databases within an organization and to apply standardized analytics tools for conducting observational research.
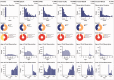
Materials and methods: Six deidentified patient-level datasets were transformed to the OMOP CDM. We evaluated the extent of information loss that occurred through the standardization process. We developed a standardized analytic tool to replicate the cohort construction process from a published epidemiology protocol and applied the analysis to all 6 databases to assess time-to-execution and comparability of results.
Results: Transformation to the CDM resulted in minimal information loss across all 6 databases. Patients and observations excluded were due to identified data quality issues in the source system, 96% to 99% of condition records and 90% to 99% of drug records were successfully mapped into the CDM using the standard vocabulary. The full cohort replication and descriptive baseline summary was executed for 2 cohorts in 6 databases in less than 1 hour.
Discussion: The standardization process improved data quality, increased efficiency, and facilitated cross-database comparisons to support a more systematic approach to observational research. Comparisons across data sources showed consistency in the impact of inclusion criteria, using the protocol and identified differences in patient characteristics and coding practices across databases.
Conclusion: Standardizing data structure (through a CDM), content (through a standard vocabulary with source code mappings), and analytics can enable an institution to apply a network-based approach to observational research across multiple, disparate observational health databases.
Keywords: controlled health services research; database; factual vocabulary; medical informatics observational study.
© The Author 2015. Published by Oxford University Press on behalf of the American Medical Informatics Association.
Figures
References
-
- Schneeweiss S, Avorn J. A review of uses of health care utilization databases for epidemiologic research on therapeutics. J Clin Epidemiol. 2005;58(4):323–337. - PubMed
-
- Madigan D, Stang PE, Berlin JA, et al. A systematic statistical approach to evaluating evidence from observational studies. Ann Rev Stat Appl. 2014;1(1):11–39.
-
- Psaty BM, Furberg CD. COX-2 inhibitors–lessons in drug safety. N Engl J Med. 2005;352 (11):1133–1135. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

