Huntingtin-associated protein 1 interacts with breakpoint cluster region protein to regulate neuronal differentiation
- PMID: 25671650
- PMCID: PMC4324908
- DOI: 10.1371/journal.pone.0116372
Huntingtin-associated protein 1 interacts with breakpoint cluster region protein to regulate neuronal differentiation
Abstract
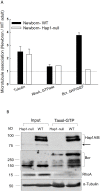
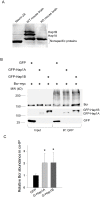
Alterations in microtubule-dependent trafficking and certain signaling pathways in neuronal cells represent critical pathogenesis in neurodegenerative diseases. Huntingtin (Htt)-associated protein-1 (Hap1) is a brain-enriched protein and plays a key role in the trafficking of neuronal surviving and differentiating cargos. Lack of Hap1 reduces signaling through tropomyosin-related kinases including extracellular signal regulated kinase (ERK), resulting in inhibition of neurite outgrowth, hypothalamic dysfunction and postnatal lethality in mice. To examine how Hap1 is involved in microtubule-dependent trafficking and neuronal differentiation, we performed a proteomic analysis using taxol-precipitated microtubules from Hap1-null and wild-type mouse brains. Breakpoint cluster region protein (Bcr), a Rho GTPase regulator, was identified as a Hap1-interacting partner. Bcr was co-immunoprecipitated with Hap1 from transfected neuro-2a cells and co-localized with Hap1A isoform more in the differentiated than in the nondifferentiated cells. The Bcr downstream effectors, namely ERK and p38, were significantly less activated in Hap1-null than in wild-type mouse hypothalamus. In conclusion, Hap1 interacts with Bcr on microtubules to regulate neuronal differentiation.
Conflict of interest statement
Figures


Similar articles
-
Interaction of Huntingtin-associated protein-1 with kinesin light chain: implications in intracellular trafficking in neurons.J Biol Chem. 2006 Feb 10;281(6):3552-9. doi: 10.1074/jbc.M509806200. Epub 2005 Dec 8. J Biol Chem. 2006. PMID: 16339760
-
Huntingtin-associated protein-1 is a synapsin I-binding protein regulating synaptic vesicle exocytosis and synapsin I trafficking.J Neurochem. 2016 Sep;138(5):710-21. doi: 10.1111/jnc.13703. Epub 2016 Jul 18. J Neurochem. 2016. PMID: 27315547
-
Huntingtin-associated protein-1 deficiency in orexin-producing neurons impairs neuronal process extension and leads to abnormal behavior in mice.J Biol Chem. 2010 May 21;285(21):15941-9. doi: 10.1074/jbc.M110.107318. Epub 2010 Mar 19. J Biol Chem. 2010. PMID: 20304926 Free PMC article.
-
Regulation of intracellular HAP1 trafficking.J Neurosci Res. 2007 Nov 1;85(14):3025-9. doi: 10.1002/jnr.21326. J Neurosci Res. 2007. PMID: 17474105 Review.
-
Huntingtin associated protein 1 and its functions.Cell Adh Migr. 2009 Jan-Mar;3(1):71-6. doi: 10.4161/cam.3.1.7511. Epub 2009 Jan 26. Cell Adh Migr. 2009. PMID: 19262167 Free PMC article. Review.
Cited by
-
Behavioral analysis of the huntingtin-associated protein 1 ortholog trak-1 in Caenorhabditis elegans.J Neurosci Res. 2016 Sep;94(9):850-6. doi: 10.1002/jnr.23756. Epub 2016 Jun 19. J Neurosci Res. 2016. PMID: 27319755 Free PMC article.
-
Research advances in huntingtin-associated protein 1 and its application prospects in diseases.Front Neurosci. 2024 Jun 21;18:1402996. doi: 10.3389/fnins.2024.1402996. eCollection 2024. Front Neurosci. 2024. PMID: 38975245 Free PMC article. Review.
-
Huntingtin-Associated Protein 1A Regulates Store-Operated Calcium Entry in Medium Spiny Neurons From Transgenic YAC128 Mice, a Model of Huntington's Disease.Front Cell Neurosci. 2018 Oct 26;12:381. doi: 10.3389/fncel.2018.00381. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30455632 Free PMC article.
-
Expression of AHI1 Rescues Amyloidogenic Pathology in Alzheimer's Disease Model Cells.Mol Neurobiol. 2019 Nov;56(11):7572-7582. doi: 10.1007/s12035-019-1587-1. Epub 2019 May 7. Mol Neurobiol. 2019. PMID: 31062249
-
Identification of the differentially expressed proteins in nasopharyngeal carcinoma by proteomics.Transl Cancer Res. 2020 Jan;9(1):21-29. doi: 10.21037/tcr.2019.11.14. Transl Cancer Res. 2020. PMID: 35117154 Free PMC article.
References
-
- Hirokawa N, Takemura R (2005) Molecular motors and mechanisms of directional transport in neurons. Nature reviews Neuroscience 6: 201–214. - PubMed
-
- Li XJ, Li SH, Sharp AH, Nucifora FC Jr, Schilling G, et al. (1995) A huntingtin-associated protein enriched in brain with implications for pathology. Nature 378: 398–402. - PubMed
-
- Rong J, Li SH, Li XJ (2007) Regulation of intracellular HAP1 trafficking. J Neurosci Res 85: 3025–3029. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

