Pemetrexed-Erlotinib, Pemetrexed Alone, or Erlotinib Alone as Second-Line Treatment for East Asian and Non-East Asian Never-Smokers with Locally Advanced or Metastatic Nonsquamous Non-small Cell Lung Cancer: Exploratory Subgroup Analysis of a Phase II Trial
- PMID: 25672577
- PMCID: PMC4614205
- DOI: 10.4143/crt.2014.051
Pemetrexed-Erlotinib, Pemetrexed Alone, or Erlotinib Alone as Second-Line Treatment for East Asian and Non-East Asian Never-Smokers with Locally Advanced or Metastatic Nonsquamous Non-small Cell Lung Cancer: Exploratory Subgroup Analysis of a Phase II Trial
Abstract
Purpose: This subgroup analysis of a phase II trial was conducted to assess possible ethnicity-based trends in efficacy and safety in East Asian (EA) and non-EA populations with nonsquamous non-small cell lung cancer (NSCLC).
Materials and methods: Never-smoker patients (n=240) with locally advanced or metastatic nonsquamous NSCLC included 133 EA patients randomized to pemetrexed supplemented with dexamethasone, folic acid, and vitamin B12 plus erlotinib (pemetrexed-erlotinib) (n=41), erlotinib (n=49), or pemetrexed (n=43), and 107 non-EA patients randomized to pemetrexed-erlotinib (n=37), erlotinib (n=33), or pemetrexed (n=37). The primary endpoint, progression-free survival (PFS), was analyzed using a multivariate Cox model.
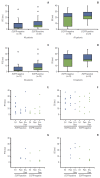
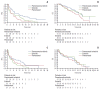
Results: Consistent with the results of the overall study, a statistically significant difference in PFS among the three arms was noted in the EA population favoring pemetrexed-erlotinib (overall p=0.003) as compared with either single-agent arm (hazard ratio [HR], 0.48; 95% confidence interval [CI], 0.29 to 0.79; p=0.004 vs. erlotinib; HR, 0.40; 95% CI, 0.23 to 0.70; p=0.001 vs. pemetrexed). The EA patients treated with pemetrexed-erlotinib achieved a longer median PFS (7.4 months) compared with erlotinib (4.5 months) and pemetrexed (4.0 months). The PFS results also numerically favored pemetrexed-erlotinib in the non-EA population (overall p=0.210) (HR, 0.62; 95% CI, 0.37 to 1.05; p=0.078 vs. erlotinib; HR, 0.75; 95% CI, 0.42 to 1.32; p=0.320 vs. pemetrexed) (median PFS: pemetrexed-erlotinib, 6.7 months; erlotinib, 3.0 months; pemetrexed, 4.4 months).
Conclusion: The PFS results from this subset analysis in both EA and non-EA populations are consistent with the results in the overall population. The PFS advantage for pemetrexed-erlotinib is significant compared with the single agents in EA patients.
Keywords: East Asian; Erlotinib; Nonsquamous non-small cell lung cancer; Pemetrexed.
Conflict of interest statement
Dae Ho Lee has received honoraria from Eli Lilly and Company as a member of advisory board. Xin Wang, Jongseok Kim, and Mauro Orlando are employees of Eli Lilly and Company, with some owning stock in Eli Lilly and Company. All other authors declare no conflicts of interest.
Figures

References
-
- Scagliotti GV, Parikh P, von Pawel J, Biesma B, Vansteenkiste J, Manegold C, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapynaive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26:3543–51. - PubMed
-
- Ciuleanu T, Brodowicz T, Zielinski C, Kim JH, Krzakowski M, Laack E, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. Lancet. 2009;374:1432–40. - PubMed
-
- Hanna N, Shepherd FA, Fossella FV, Pereira JR, De Marinis F, von Pawel J, et al. Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol. 2004;22:1589–97. - PubMed
-
- Yang CH, Simms L, Park K, Lee JS, Scagliotti G, Orlando M. Efficacy and safety of cisplatin/pemetrexed versus cisplatin/gemcitabine as first-line treatment in East Asian patients with advanced non-small cell lung cancer: results of an exploratory subgroup analysis of a phase III trial. J Thorac Oncol. 2010;5:688–95. - PubMed
-
- Sun Y, Wu YL, Zhou CC, Zhang L, Zhang L, Liu XY, et al. Second-line pemetrexed versus docetaxel in Chinese patients with locally advanced or metastatic non-small cell lung cancer: a randomized, open-label study. Lung Cancer. 2013;79:143–50. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

