Characteristic patterns of dendritic remodeling in early-stage glaucoma: evidence from genetically identified retinal ganglion cell types
- PMID: 25673829
- PMCID: PMC6605614
- DOI: 10.1523/JNEUROSCI.1419-14.2015
Characteristic patterns of dendritic remodeling in early-stage glaucoma: evidence from genetically identified retinal ganglion cell types
Abstract
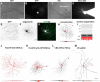
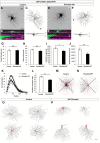
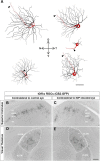
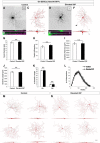
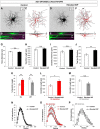
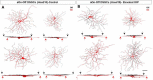
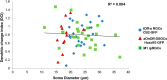
Retinal ganglion cell (RGC) loss is a hallmark of glaucoma and the second leading cause of blindness worldwide. The type and timing of cellular changes leading to RGC loss in glaucoma remain incompletely understood, including whether specific RGC subtypes are preferentially impacted at early stages of this disease. Here we applied the microbead occlusion model of glaucoma to different transgenic mouse lines, each expressing green fluorescent protein in 1-2 specific RGC subtypes. Targeted filling, reconstruction, and subsequent comparison of the genetically identified RGCs in control and bead-injected eyes revealed that some subtypes undergo significant dendritic rearrangements as early as 7 d following induction of elevated intraocular pressure (IOP). By comparing specific On-type, On-Off-type and Off-type RGCs, we found that RGCs that target the majority of their dendritic arbors to the scleral half or "Off" sublamina of the inner plexiform layer (IPL) undergo the greatest changes, whereas RGCs with the majority of their dendrites in the On sublamina did not alter their structure at this time point. Moreover, M1 intrinsically photosensitive RGCs, which functionally are On RGCs but structurally stratify their dendrites in the Off sublamina of the IPL, also underwent significant changes in dendritic structure 1 week after elevated IOP. Thus, our findings reveal that certain RGC subtypes manifest significant changes in dendritic structure after very brief exposure to elevated IOP. The observation that RGCs stratifying most of their dendrites in the Off sublamina are first to alter their structure may inform the development of new strategies to detect, monitor, and treat glaucoma in humans.
Keywords: dendrites; glaucoma; retinal ganglion cells.
Copyright © 2015 the authors 0270-6474/15/352329-15$15.00/0.
Figures



References
-
- Berson DM. Retinal ganglion cell types and their central projection. In: Masland R, editor. The senses: a comprehensive reference. Oxford: Elsevier; 2008.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
