The JAK/STAT3 pathway is a common inducer of astrocyte reactivity in Alzheimer's and Huntington's diseases
- PMID: 25673868
- PMCID: PMC6605603
- DOI: 10.1523/JNEUROSCI.3516-14.2015
The JAK/STAT3 pathway is a common inducer of astrocyte reactivity in Alzheimer's and Huntington's diseases
Abstract
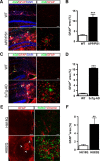
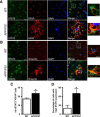
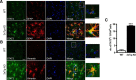
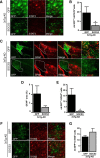
Astrocyte reactivity is a hallmark of neurodegenerative diseases (ND), but its effects on disease outcomes remain highly debated. Elucidation of the signaling cascades inducing reactivity in astrocytes during ND would help characterize the function of these cells and identify novel molecular targets to modulate disease progression. The Janus kinase/signal transducer and activator of transcription 3 (JAK/STAT3) pathway is associated with reactive astrocytes in models of acute injury, but it is unknown whether this pathway is directly responsible for astrocyte reactivity in progressive pathological conditions such as ND. In this study, we examined whether the JAK/STAT3 pathway promotes astrocyte reactivity in several animal models of ND. The JAK/STAT3 pathway was activated in reactive astrocytes in two transgenic mouse models of Alzheimer's disease and in a mouse and a nonhuman primate lentiviral vector-based model of Huntington's disease (HD). To determine whether this cascade was instrumental for astrocyte reactivity, we used a lentiviral vector that specifically targets astrocytes in vivo to overexpress the endogenous inhibitor of the JAK/STAT3 pathway [suppressor of cytokine signaling 3 (SOCS3)]. SOCS3 significantly inhibited this pathway in astrocytes, prevented astrocyte reactivity, and decreased microglial activation in models of both diseases. Inhibition of the JAK/STAT3 pathway within reactive astrocytes also increased the number of huntingtin aggregates, a neuropathological hallmark of HD, but did not influence neuronal death. Our data demonstrate that the JAK/STAT3 pathway is a common mediator of astrocyte reactivity that is highly conserved between disease states, species, and brain regions. This universal signaling cascade represents a potent target to study the role of reactive astrocytes in ND.
Keywords: SOCS3; STAT3; animal models; lentiviral vector; neurodegenerative diseases; reactive astrocytes.
Copyright © 2015 the authors 0270-6474/15/352817-13$15.00/0.
Figures





Similar articles
-
Modulation of astrocyte reactivity improves functional deficits in mouse models of Alzheimer's disease.Acta Neuropathol Commun. 2018 Oct 16;6(1):104. doi: 10.1186/s40478-018-0606-1. Acta Neuropathol Commun. 2018. PMID: 30322407 Free PMC article.
-
The complex STATes of astrocyte reactivity: How are they controlled by the JAK-STAT3 pathway?Neuroscience. 2016 Aug 25;330:205-18. doi: 10.1016/j.neuroscience.2016.05.043. Epub 2016 May 27. Neuroscience. 2016. PMID: 27241943 Review.
-
Microglial Janus kinase/signal transduction and activator of transcription 3 pathway activity directly impacts astrocyte and spinal neuron characteristics.J Neurochem. 2016 Jan;136(1):133-47. doi: 10.1111/jnc.13375. Epub 2015 Oct 28. J Neurochem. 2016. PMID: 26440453
-
Activation of the JAK-STAT3 pathway is associated with the growth of colorectal carcinoma cells.Oncol Rep. 2014 Jan;31(1):335-41. doi: 10.3892/or.2013.2858. Epub 2013 Nov 20. Oncol Rep. 2014. PMID: 24253664
-
The roles of SOCS3 and STAT3 in bacterial infection and inflammatory diseases.Scand J Immunol. 2018 Dec;88(6):e12727. doi: 10.1111/sji.12727. Scand J Immunol. 2018. PMID: 30341772 Review.
Cited by
-
Neuromodulation of Glial Function During Neurodegeneration.Front Cell Neurosci. 2020 Aug 21;14:278. doi: 10.3389/fncel.2020.00278. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32973460 Free PMC article.
-
Role of glia in optic nerve.Prog Retin Eye Res. 2021 Mar;81:100886. doi: 10.1016/j.preteyeres.2020.100886. Epub 2020 Aug 6. Prog Retin Eye Res. 2021. PMID: 32771538 Free PMC article. Review.
-
SRF-deficient astrocytes provide neuroprotection in mouse models of excitotoxicity and neurodegeneration.Elife. 2024 Feb 9;13:e95577. doi: 10.7554/eLife.95577. Elife. 2024. PMID: 38289036 Free PMC article.
-
Megalencephalic Leukoencephalopathy with Subcortical Cysts Protein-1 (MLC1) Counteracts Astrocyte Activation in Response to Inflammatory Signals.Mol Neurobiol. 2019 Dec;56(12):8237-8254. doi: 10.1007/s12035-019-01657-y. Epub 2019 Jun 17. Mol Neurobiol. 2019. PMID: 31209783
-
Stat3 mediates Fyn kinase-driven dopaminergic neurodegeneration and microglia activation.Dis Model Mech. 2024 Dec 1;17(12):dmm052011. doi: 10.1242/dmm.052011. Epub 2024 Dec 6. Dis Model Mech. 2024. PMID: 39641161 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
