Recurrent gain-of-function USP8 mutations in Cushing's disease
- PMID: 25675982
- PMCID: PMC4349249
- DOI: 10.1038/cr.2015.20
Recurrent gain-of-function USP8 mutations in Cushing's disease
Abstract
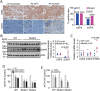
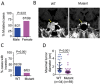
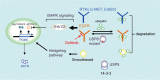
Cushing's disease, also known as adrenocorticotropic hormone (ACTH)-secreting pituitary adenomas (PAs) that cause excess cortisol production, accounts for up to 85% of corticotrophin-dependent Cushing's syndrome cases. However, the genetic alterations in this disease are unclear. Here, we performed whole-exome sequencing of DNA derived from 12 ACTH-secreting PAs and matched blood samples, which revealed three types of somatic mutations in a candidate gene, USP8 (encoding ubiquitin-specific protease 8), exclusively in exon 14 in 8 of 12 ACTH-secreting PAs. We further evaluated somatic USP8 mutations in additional 258 PAs by Sanger sequencing. Targeted sequencing further identified a total of 17 types of USP8 variants in 67 of 108 ACTH-secreting PAs (62.04%). However, none of these mutations was detected in other types of PAs (n = 150). These mutations aggregate within the 14-3-3 binding motif of USP8 and disrupt the interaction between USP8 and 14-3-3 protein, resulting in an elevated capacity to protect EGFR from lysosomal degradation. Accordingly, PAs with mutated USP8 display a higher incidence of EGFR expression, elevated EGFR protein abundance and mRNA expression levels of POMC, which encodes the precursor of ACTH. PAs with mutated USP8 are significantly smaller in size and have higher ACTH production than wild-type PAs. In surgically resected primary USP8-mutated tumor cells, USP8 knockdown or blocking EGFR effectively attenuates ACTH secretion. Taken together, somatic gain-of-function USP8 mutations are common and contribute to ACTH overproduction in Cushing's disease. Inhibition of USP8 or EGFR is promising for treating USP8-mutated corticotrophin adenoma. Our study highlights the potentially functional mutated gene in Cushing's disease and provides insights into the therapeutics of this disease.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

