Ab initio dynamics of the cytochrome P450 hydroxylation reaction
- PMID: 25681906
- PMCID: PMC4367892
- DOI: 10.1063/1.4907733
Ab initio dynamics of the cytochrome P450 hydroxylation reaction
Abstract
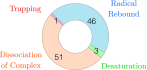
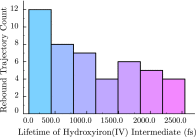
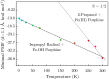
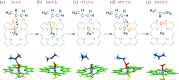
The iron(IV)-oxo porphyrin π-cation radical known as Compound I is the primary oxidant within the cytochromes P450, allowing these enzymes to affect the substrate hydroxylation. In the course of this reaction, a hydrogen atom is abstracted from the substrate to generate hydroxyiron(IV) porphyrin and a substrate-centered radical. The hydroxy radical then rebounds from the iron to the substrate, yielding the hydroxylated product. While Compound I has succumbed to theoretical and spectroscopic characterization, the associated hydroxyiron species is elusive as a consequence of its very short lifetime, for which there are no quantitative estimates. To ascertain the physical mechanism underlying substrate hydroxylation and probe this timescale, ab initio molecular dynamics simulations and free energy calculations are performed for a model of Compound I catalysis. Semiclassical estimates based on these calculations reveal the hydrogen atom abstraction step to be extremely fast, kinetically comparable to enzymes such as carbonic anhydrase. Using an ensemble of ab initio simulations, the resultant hydroxyiron species is found to have a similarly short lifetime, ranging between 300 fs and 3600 fs, putatively depending on the enzyme active site architecture. The addition of tunneling corrections to these rates suggests a strong contribution from nuclear quantum effects, which should accelerate every step of substrate hydroxylation by an order of magnitude. These observations have strong implications for the detection of individual hydroxylation intermediates during P450 catalysis.
Figures
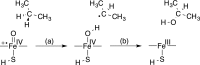


Similar articles
-
Effect of the axial ligand on substrate sulfoxidation mediated by iron(IV)-oxo porphyrin cation radical oxidants.Chemistry. 2011 May 23;17(22):6196-205. doi: 10.1002/chem.201003187. Epub 2011 Apr 5. Chemistry. 2011. PMID: 21469227
-
Radical intermediates in the catalytic oxidation of hydrocarbons by bacterial and human cytochrome P450 enzymes.Biochemistry. 2006 Jan 17;45(2):533-42. doi: 10.1021/bi051840z. Biochemistry. 2006. PMID: 16401082 Free PMC article.
-
Bioengineering of Cytochrome P450 OleTJE: How Does Substrate Positioning Affect the Product Distributions?Molecules. 2020 Jun 9;25(11):2675. doi: 10.3390/molecules25112675. Molecules. 2020. PMID: 32526971 Free PMC article.
-
One oxidant, many pathways: a theoretical perspective of monooxygenation mechanisms by cytochrome P450 enzymes.J Biol Inorg Chem. 2004 Sep;9(6):661-8. doi: 10.1007/s00775-004-0576-6. Epub 2004 Jul 28. J Biol Inorg Chem. 2004. PMID: 15365903 Review.
-
Alternatives to the oxoferryl porphyrin cation radical as the proposed reactive intermediate of cytochrome P450: two-electron oxidized Fe(III) porphyrin derivatives.J Biol Inorg Chem. 2001 Oct;6(8):846-56. doi: 10.1007/s007750100278. J Biol Inorg Chem. 2001. PMID: 11713692 Review.
Cited by
-
Mechanism of Melatonin Metabolism by CYP1A1: What Determines the Bifurcation Pathways of Hydroxylation versus Deformylation?J Phys Chem B. 2022 Nov 24;126(46):9591-9606. doi: 10.1021/acs.jpcb.2c07200. Epub 2022 Nov 15. J Phys Chem B. 2022. PMID: 36380557 Free PMC article.
-
Mapping hole hopping escape routes in proteins.Proc Natl Acad Sci U S A. 2019 Aug 6;116(32):15811-15816. doi: 10.1073/pnas.1906394116. Epub 2019 Jul 24. Proc Natl Acad Sci U S A. 2019. PMID: 31341081 Free PMC article.
-
Mechanism of the Oxidative Ring-Closure Reaction during Gliotoxin Biosynthesis by Cytochrome P450 GliF.Int J Mol Sci. 2024 Aug 6;25(16):8567. doi: 10.3390/ijms25168567. Int J Mol Sci. 2024. PMID: 39201254 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

