Phosphate uptake-independent signaling functions of the type III sodium-dependent phosphate transporter, PiT-1, in vascular smooth muscle cells
- PMID: 25684711
- PMCID: PMC4387109
- DOI: 10.1016/j.yexcr.2015.02.002
Phosphate uptake-independent signaling functions of the type III sodium-dependent phosphate transporter, PiT-1, in vascular smooth muscle cells
Abstract
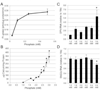
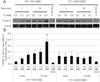
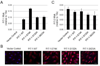
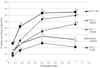
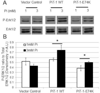
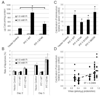
Vascular calcification (VC) is prevalent in chronic kidney disease and elevated serum inorganic phosphate (Pi) is a recognized risk factor. The type III sodium-dependent phosphate transporter, PiT-1, is required for elevated Pi-induced osteochondrogenic differentiation and matrix mineralization in vascular smooth muscle cells (VSMCs). However, the molecular mechanism(s) by which PiT-1 promotes these processes is unclear. In the present study, we confirmed that the Pi concentration required to induce osteochondrogenic differentiation and matrix mineralization of mouse VSMCs was well above that required for maximal Pi uptake, suggesting a signaling function of PiT-1 that was independent of Pi transport. Elevated Pi-induced signaling via ERK1/2 phosphorylation was abrogated in PiT-1 deficient VSMCs, but could be rescued by wild-type (WT) and a Pi transport-deficient PiT-1 mutant. Furthermore, both WT and transport-deficient PiT-1 mutants promoted osteochondrogenic differentiation as measured by decreased SM22α and increased osteopontin mRNA expression. Finally, compared to vector alone, expression of transport-deficient PiT-1 mutants promoted VSMC matrix mineralization, but not to the extent observed with PiT-1 WT. These data suggest that both Pi uptake-dependent and -independent functions of PiT-1 are important for VSMC processes mediating vascular calcification.
Keywords: Calcification; ERK phosphorylation; Phosphate; PiT-1; SLC20A1; Signaling.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Adapter Protein RapGEF1 Is Required for ERK1/2 Signaling in Response to Elevated Phosphate in Vascular Smooth Muscle Cells.J Vasc Res. 2021;58(5):277-285. doi: 10.1159/000516044. Epub 2021 May 5. J Vasc Res. 2021. PMID: 33951626 Free PMC article.
-
TDAG51 (T-Cell Death-Associated Gene 51) Is a Key Modulator of Vascular Calcification and Osteogenic Transdifferentiation of Arterial Smooth Muscle Cells.Arterioscler Thromb Vasc Biol. 2020 Jul;40(7):1664-1679. doi: 10.1161/ATVBAHA.119.313779. Epub 2020 May 21. Arterioscler Thromb Vasc Biol. 2020. PMID: 32434409
-
Oxidative stress by Ca2+ overload is critical for phosphate-induced vascular calcification.Am J Physiol Heart Circ Physiol. 2020 Dec 1;319(6):H1302-H1312. doi: 10.1152/ajpheart.00305.2020. Epub 2020 Oct 23. Am J Physiol Heart Circ Physiol. 2020. PMID: 33095057
-
Sodium-dependent phosphate cotransporters and vascular calcification.Curr Opin Nephrol Hypertens. 2007 Jul;16(4):325-8. doi: 10.1097/MNH.0b013e3281c55ef1. Curr Opin Nephrol Hypertens. 2007. PMID: 17565274 Review.
-
Extracellular phosphate as a signaling molecule.Contrib Nephrol. 2013;180:14-24. doi: 10.1159/000346776. Epub 2013 May 3. Contrib Nephrol. 2013. PMID: 23652547 Review.
Cited by
-
Matrix Vesicles: Role in Bone Mineralization and Potential Use as Therapeutics.Pharmaceuticals (Basel). 2021 Mar 24;14(4):289. doi: 10.3390/ph14040289. Pharmaceuticals (Basel). 2021. PMID: 33805145 Free PMC article. Review.
-
The Complexities of Organ Crosstalk in Phosphate Homeostasis: Time to Put Phosphate Sensing Back in the Limelight.Int J Mol Sci. 2021 May 27;22(11):5701. doi: 10.3390/ijms22115701. Int J Mol Sci. 2021. PMID: 34071837 Free PMC article. Review.
-
FGF23 and Phosphate-Cardiovascular Toxins in CKD.Toxins (Basel). 2019 Nov 6;11(11):647. doi: 10.3390/toxins11110647. Toxins (Basel). 2019. PMID: 31698866 Free PMC article. Review.
-
Phosphate Is a Cardiovascular Toxin.Adv Exp Med Biol. 2022;1362:107-134. doi: 10.1007/978-3-030-91623-7_11. Adv Exp Med Biol. 2022. PMID: 35288877 Review.
-
Oxidative Stress Related to Plasmalemmal and Mitochondrial Phosphate Transporters in Vascular Calcification.Antioxidants (Basel). 2022 Mar 2;11(3):494. doi: 10.3390/antiox11030494. Antioxidants (Basel). 2022. PMID: 35326144 Free PMC article. Review.
References
-
- Detrano R, Guerci AD, Carr JJ, Bild DE, Burke G, Folsom AR, Liu K, Shea S, Szklo M, Bluemke DA, O'Leary DH, Tracy R, Watson K, Wong ND, Kronmal RA. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med. 2008;358:1336–1345. - PubMed
-
- Shroff RC, McNair R, Figg N, Skepper JN, Schurgers L, Gupta A, Hiorns M, Donald AE, Deanfield J, Rees L, Shanahan CM. Dialysis accelerates medial vascular calcification in part by triggering smooth muscle cell apoptosis. Circulation. 2008;118:1748–1757. - PubMed
-
- Tonelli M, Wiebe N, Culleton B, House A, Rabbat C, Fok M, McAlister F, Garg AX. Chronic kidney disease and mortality risk: a systematic review. J Am Soc Nephrol. 2006;17:2034–2047. - PubMed
-
- Block GA, Hulbert-Shearon TE, Levin NW, Port FK. Association of serum phosphorus and calcium × phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am J Kidney Dis. 1998;31:607–617. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

