Biomineralized matrices dominate soluble cues to direct osteogenic differentiation of human mesenchymal stem cells through adenosine signaling
- PMID: 25686297
- PMCID: PMC6277021
- DOI: 10.1021/acs.biomac.5b00099
Biomineralized matrices dominate soluble cues to direct osteogenic differentiation of human mesenchymal stem cells through adenosine signaling
Abstract
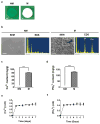
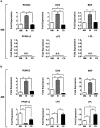
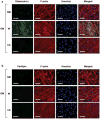
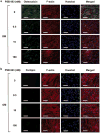
Stem cell differentiation is determined by a repertoire of signals from its microenvironment, which includes the extracellular matrix (ECM) and soluble cues. The ability of mesenchymal stem cells (MSCs), a common precursor for the skeletal system, to differentiate into osteoblasts and adipocytes in response to their local cues plays an important role in skeletal tissue regeneration and homeostasis. In this study, we investigated whether a bone-specific calcium phosphate (CaP) mineral environment could induce osteogenic differentiation of human MSCs, while inhibiting their adipogenic differentiation, in the presence of adipogenic-inducing medium. We also examined the mechanism through which the mineralized matrix suppresses adipogenesis of hMSCs to promote their osteogenic differentiation. Our results show that hMSCs cultured on mineralized matrices underwent osteogenic differentiation despite being cultured in the presence of adipogenic medium, which indicates the dominance of matrix-based cues of the mineralized matrix in directing osteogenic commitment of stem cells. Furthermore, the mineralized matrix-driven attenuation of adipogenesis was reversed with the inhibition of A2b adenosine receptor (A2bR), implicating a role of adenosine signaling in mineralized environment-mediated inhibition of adipogenesis. Such synthetic matrices with an intrinsic ability to direct differentiation of multipotent adult stem cells toward a targeted phenotype while inhibiting their differentiation into other lineages not only will be a powerful tool in delineating the role of complex microenvironmental cues on stem cell commitment but also will contribute to functional tissue engineering and their translational applications.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Deans RJ, Moseley AB. Mesenchymal stem cells: biology and potential clinical uses. Exp Hematol. 2000;28:875–884. - PubMed
-
- Hwang NS, Zhang C, Hwang YS, Varghese S. Mesenchymal stem cell differentiation and roles in regenerative medicine. Wiley Interdiscip Rev: Syst Biol Med. 2009;1:97–106. - PubMed
-
- Beresford J, Bennett J, Devlin C, Leboy P, Owen M. Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J Cell Sci. 1992;102:341–351. - PubMed
-
- Jaiswal RK, Jaiswal N, Bruder SP, Mbalaviele G, Marshak DR, Pittenger MF. Adult human mesenchymal stem cell differentiation to the osteogenic or adipogenic lineage is regulated by mitogen-activated protein kinase. J Biol Chem. 2000;275:9645–9652. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

