Titania nanotube arrays as interfaces for neural prostheses
- PMID: 25687003
- PMCID: PMC4331648
- DOI: 10.1016/j.msec.2015.01.077
Titania nanotube arrays as interfaces for neural prostheses
Abstract
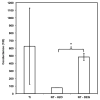
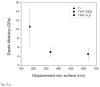
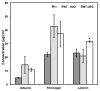
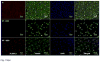
Neural prostheses have become ever more acceptable treatments for many different types of neurological damage and disease. Here we investigate the use of two different morphologies of titania nanotube arrays as interfaces to advance the longevity and effectiveness of these prostheses. The nanotube arrays were characterized for their nanotopography, crystallinity, conductivity, wettability, surface mechanical properties and adsorption of key proteins: fibrinogen, albumin and laminin. The loosely packed nanotube arrays fabricated using a diethylene glycol based electrolyte, contained a higher presence of the anatase crystal phase and were subsequently more conductive. These arrays yielded surfaces with higher wettability and lower modulus than the densely packed nanotube arrays fabricated using water based electrolyte. Further the adhesion, proliferation and differentiation of the C17.2 neural stem cell line was investigated on the nanotube arrays. The proliferation ratio of the cells as well as the level of neuronal differentiation was seen to increase on the loosely packed arrays. The results indicate that loosely packed nanotube arrays similar to the ones produced here with a DEG based electrolyte, may provide a favorable template for growth and maintenance of C17.2 neural stem cell line.
Keywords: Astrocytes; C17.2 neural stem cell line; Neural prostheses; Neurons; Titania nanotube arrays.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures













References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

