Distinct roles of the DmNav and DSC1 channels in the action of DDT and pyrethroids
- PMID: 25687544
- PMCID: PMC4486006
- DOI: 10.1016/j.neuro.2015.02.001
Distinct roles of the DmNav and DSC1 channels in the action of DDT and pyrethroids
Abstract
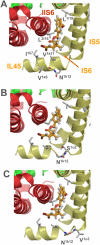
Voltage-gated sodium channels (Nav channels) are critical for electrical signaling in the nervous system and are the primary targets of the insecticides DDT and pyrethroids. In Drosophila melanogaster, besides the canonical Nav channel, Para (also called DmNav), there is a sodium channel-like cation channel called DSC1 (Drosophila sodium channel 1). Temperature-sensitive paralytic mutations in DmNav (para(ts)) confer resistance to DDT and pyrethroids, whereas DSC1 knockout flies exhibit enhanced sensitivity to pyrethroids. To further define the roles and interaction of DmNav and DSC1 channels in DDT and pyrethroid neurotoxicology, we generated a DmNav/DSC1 double mutant line by introducing a para(ts1) allele (carrying the I265N mutation) into a DSC1 knockout line. We confirmed that the I265N mutation reduced the sensitivity to two pyrethroids, permethrin and deltamethrin of a DmNav variant expressed in Xenopus oocytes. Computer modeling predicts that the I265N mutation confers pyrethroid resistance by allosterically altering the second pyrethroid receptor site on the DmNav channel. Furthermore, we found that I265N-mediated pyrethroid resistance in para(ts1) mutant flies was almost completely abolished in para(ts1);DSC1(-/-) double mutant flies. Unexpectedly, however, the DSC1 knockout flies were less sensitive to DDT, compared to the control flies (w(1118A)), and the para(ts1);DSC1(-/-) double mutant flies were even more resistant to DDT compared to the DSC1 knockout or para(ts1) mutant. Our findings revealed distinct roles of the DmNav and DSC1 channels in the neurotoxicology of DDT vs. pyrethroids and implicate the exciting possibility of using DSC1 channel blockers or modifiers in the management of pyrethroid resistance.
Keywords: DDT; DSC1; Pyrethroids insecticide resistance; Voltage-gated sodium channel.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
DSC1 channel-dependent developmental regulation of pyrethroid susceptibility in Drosophila melanogaster.Pestic Biochem Physiol. 2018 Jun;148:190-198. doi: 10.1016/j.pestbp.2018.04.014. Epub 2018 Apr 27. Pestic Biochem Physiol. 2018. PMID: 29891372
-
Negative cross-resistance of a pyrethroid-resistant Drosophila mutant to Phryma leptostachya-derived haedoxan A.Insect Sci. 2022 Jun;29(3):817-826. doi: 10.1111/1744-7917.12973. Epub 2021 Oct 25. Insect Sci. 2022. PMID: 34547832
-
Molecular evidence of sequential evolution of DDT- and pyrethroid-resistant sodium channel in Aedes aegypti.PLoS Negl Trop Dis. 2019 Jun 3;13(6):e0007432. doi: 10.1371/journal.pntd.0007432. eCollection 2019 Jun. PLoS Negl Trop Dis. 2019. PMID: 31158225 Free PMC article.
-
The molecular interactions of pyrethroid insecticides with insect and mammalian sodium channels.Pest Manag Sci. 2001 Oct;57(10):877-88. doi: 10.1002/ps.392. Pest Manag Sci. 2001. PMID: 11695180 Review.
-
The Drosophila Sodium Channel 1 (DSC1): The founding member of a new family of voltage-gated cation channels.Pestic Biochem Physiol. 2015 May;120:36-9. doi: 10.1016/j.pestbp.2014.12.005. Epub 2014 Dec 9. Pestic Biochem Physiol. 2015. PMID: 25987218 Review.
Cited by
-
Signatures of Insecticide Selection in the Genome of Drosophila melanogaster.G3 (Bethesda). 2018 Nov 6;8(11):3469-3480. doi: 10.1534/g3.118.200537. G3 (Bethesda). 2018. PMID: 30190420 Free PMC article.
-
Elucidating the kinetics and mechanisms of tetramethrin biodegradation by the fungal strain Neocosmospora sp. AF3.Microb Cell Fact. 2025 May 27;24(1):124. doi: 10.1186/s12934-025-02747-4. Microb Cell Fact. 2025. PMID: 40426141 Free PMC article.
-
Honeybee CaV4 has distinct permeation, inactivation, and pharmacology from homologous NaV channels.J Gen Physiol. 2024 May 6;156(5):e202313509. doi: 10.1085/jgp.202313509. Epub 2024 Apr 1. J Gen Physiol. 2024. PMID: 38557788 Free PMC article.
-
Evidence for Dual Binding Sites for 1,1,1-Trichloro-2,2-bis(p-chlorophenyl)ethane (DDT) in Insect Sodium Channels.J Biol Chem. 2016 Feb 26;291(9):4638-48. doi: 10.1074/jbc.M115.678672. Epub 2015 Dec 4. J Biol Chem. 2016. PMID: 26637352 Free PMC article.
-
Alanine to valine substitutions in the pore helix IIIP1 and linker-helix IIIL45 confer cockroach sodium channel resistance to DDT and pyrethroids.Neurotoxicology. 2017 May;60:197-206. doi: 10.1016/j.neuro.2016.06.009. Epub 2016 Jun 18. Neurotoxicology. 2017. PMID: 27328896 Free PMC article.
References
-
- Amichot M, Castella C, Cuany A, Berge JB, Pauron D. Target modification as a molecular mechanism of pyrethroid resistance in Drosophila melanogaster. Pestic. Biochem. Physiol. 1992;44:183–190.
-
- Beeman RW. Recent advances in mode of action of insecticides. Annu. Rev. Entomol. 1982;27:253–281. - PubMed
-
- Bloomquist JR, Mittler TE, Radovsky FJ, Resh VH. Ann. Rev. Entomol. Annual Reviews, Inc.; Palo Alto: 1996. Ion Channels as Targets for Insecticides; pp. 163–190. - PubMed
-
- Bloomquist JR, Soderlund DM. Pyrethroid insecticides and DDT modify alkaloid-dependent sodium channel activation and its enhancement by sea anemone toxin. Molec. Pharmacol. 1988;33:543–550. - PubMed
-
- Burton MJ, Mellor IR, Duce IR, Davies TGE, Field LM, Williamson MS. Differential resistance of insect sodium channels with kdr mutations to deltamethrin, permethrin and DDT. Insect Biochem. Mol. Biol. 2011;41:723–732. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

