Rapidly progressive Alzheimer's disease features distinct structures of amyloid-β
- PMID: 25688081
- PMCID: PMC5014074
- DOI: 10.1093/brain/awv006
Rapidly progressive Alzheimer's disease features distinct structures of amyloid-β
Abstract
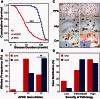
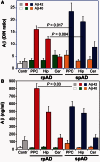
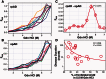
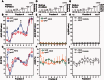
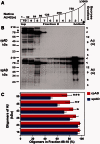
Genetic and environmental factors that increase the risk of late-onset Alzheimer disease are now well recognized but the cause of variable progression rates and phenotypes of sporadic Alzheimer's disease is largely unknown. We aimed to investigate the relationship between diverse structural assemblies of amyloid-β and rates of clinical decline in Alzheimer's disease. Using novel biophysical methods, we analysed levels, particle size, and conformational characteristics of amyloid-β in the posterior cingulate cortex, hippocampus and cerebellum of 48 cases of Alzheimer's disease with distinctly different disease durations, and correlated the data with APOE gene polymorphism. In both hippocampus and posterior cingulate cortex we identified an extensive array of distinct amyloid-β42 particles that differ in size, display of N-terminal and C-terminal domains, and conformational stability. In contrast, amyloid-β40 present at low levels did not form a major particle with discernible size, and both N-terminal and C- terminal domains were largely exposed. Rapidly progressive Alzheimer's disease that is associated with a low frequency of APOE e4 allele demonstrates considerably expanded conformational heterogeneity of amyloid-β42, with higher levels of distinctly structured amyloid-β42 particles composed of 30-100 monomers, and fewer particles composed of < 30 monomers. The link between rapid clinical decline and levels of amyloid-β42 with distinct structural characteristics suggests that different conformers may play an important role in the pathogenesis of distinct Alzheimer's disease phenotypes. These findings indicate that Alzheimer's disease exhibits a wide spectrum of amyloid-β42 structural states and imply the existence of prion-like conformational strains.
Keywords: Alzheimer; progression rate; structure; β-amyloid.
© The Author (2015). Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures
References
-
- Akiyama H, Mori H, Saido T, Kondo H, Ikeda K, McGeer PL. Occurrence of the diffuse amyloid beta-protein (Abeta) deposits with numerous Abeta-containing glial cells in the cerebral cortex of patients with Alzheimer’s disease. Glia. 1999;25:324–31. - PubMed
-
- Amieva H, Mokri H, Le Goff M, Meillon C, Jacqmin-Gadda H, Foubert-Samier A, et al. Compensatory mechanisms in higher-educated subjects with Alzheimer’s disease: a study of 20 years of cognitive decline. Brain. 2014;137(Pt 4):1167–75. - PubMed
-
- Beekly DL, Ramos EM, Lee WW, Deitrich WD, Jacka ME, Wu J, et al. The National Alzheimer’s Coordinating Center (NACC) database: the Uniform Data Set. Alzheimer Dis Assoc Disord. 2007;21:249–58. - PubMed
-
- Bellon A, Seyfert-Brandt W, Lang W, Baron H, Groner A, Vey M. Improved conformation-dependent immunoassay: suitability for human prion detection with enhanced sensitivity. J Gen Virol. 2003;84:1921–5. - PubMed
-
- Benilova I, Karran E, De Strooper B. The toxic Abeta oligomer and Alzheimer’s disease: an emperor in need of clothes. Nat Neurosci. 2012;15:349–57. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- RC2 HL102923/HL/NHLBI NIH HHS/United States
- UC2 HL102926/HL/NHLBI NIH HHS/United States
- UC2 HL103010/HL/NHLBI NIH HHS/United States
- HL-103010/HL/NHLBI NIH HHS/United States
- RC2 HL102926/HL/NHLBI NIH HHS/United States
- NS074317/NS/NINDS NIH HHS/United States
- U01AG016976/AG/NIA NIH HHS/United States
- U01 AG016976/AG/NIA NIH HHS/United States
- UR8/CCU515004/PHS HHS/United States
- HL-102924/HL/NHLBI NIH HHS/United States
- RC2 HL102924/HL/NHLBI NIH HHS/United States
- HL-102926/HL/NHLBI NIH HHS/United States
- UC2 HL102923/HL/NHLBI NIH HHS/United States
- R01 NS074317/NS/NINDS NIH HHS/United States
- UC2 HL102924/HL/NHLBI NIH HHS/United States
- HL-102925/HL/NHLBI NIH HHS/United States
- RC2 HL103010/HL/NHLBI NIH HHS/United States
- HL-102923/HL/NHLBI NIH HHS/United States
- RC2 HL102925/HL/NHLBI NIH HHS/United States
- UC2 HL102925/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

