Microbiota-Independent Ameliorative Effects of Antibiotics on Spontaneous Th2-Associated Pathology of the Small Intestine
- PMID: 25689829
- PMCID: PMC4331505
- DOI: 10.1371/journal.pone.0118795
Microbiota-Independent Ameliorative Effects of Antibiotics on Spontaneous Th2-Associated Pathology of the Small Intestine
Erratum in
-
Correction: Microbiota-Independent Ameliorative Effects of Antibiotics on Spontaneous Th2-Associated Pathology of the Small Intestine.PLoS One. 2015 Jul 20;10(7):e0133787. doi: 10.1371/journal.pone.0133787. eCollection 2015. PLoS One. 2015. PMID: 26193662 Free PMC article. No abstract available.
Abstract
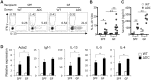
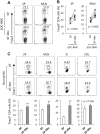
We have previously generated a mouse model of spontaneous Th2-associated disease of the small intestine called TRAF6ΔDC, in which dendritic cell (DC)-intrinsic expression of the signaling mediator TRAF6 is ablated. Interestingly, broad-spectrum antibiotic treatment ameliorates TRAF6ΔDC disease, implying a role for commensal microbiota in disease development. However, the relationship between the drug effects and commensal microbiota status remains to be formally demonstrated. To directly assess this relationship, we have now generated TRAF6ΔDC bone marrow chimera mice under germ-free (GF) conditions lacking commensal microbiota, and found, unexpectedly, that Th2-associated disease is actually exacerbated in GF TRAF6ΔDC mice compared to specific pathogen-free (SPF) TRAF6ΔDC mice. At the same time, broad-spectrum antibiotic treatment of GF TRAF6ΔDC mice has an ameliorative effect similar to that observed in antibiotics-treated SPF TRAF6ΔDC mice, implying a commensal microbiota-independent effect of broad-spectrum antibiotic treatment. We further found that treatment of GF TRAF6ΔDC mice with broad-spectrum antibiotics increases Foxp3+ Treg populations in lymphoid organs and the small intestine, pointing to a possible mechanism by which treatment may directly exert an immunomodulatory effect. To investigate links between the exacerbated phenotype of the small intestines of GF TRAF6ΔDC mice and local microbiota, we performed microbiotic profiling of the luminal contents specifically within the small intestines of diseased TRAF6ΔDC mice, and, when compared to co-housed control mice, found significantly increased total bacterial content characterized by specific increases in Firmicutes Lactobacillus species. These data suggest a protective effect of Firmicutes Lactobacillus against the spontaneous Th2-related inflammation of the small intestine of the TRAF6ΔDC model, and may represent a potential mechanism for related disease phenotypes.
Conflict of interest statement
Figures



References
-
- Morrissey PJ, Charrier K. Induction of wasting disease in SCID mice by the transfer of normal CD4+/CD45RBhi T cells and the regulation of this autoreactivity by CD4+/CD45RBlo T cells. Res Immunol. 1994;145: 357–362. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

