Avibactam and inhibitor-resistant SHV β-lactamases
- PMID: 25691639
- PMCID: PMC4468741
- DOI: 10.1128/AAC.04405-14
Avibactam and inhibitor-resistant SHV β-lactamases
Abstract
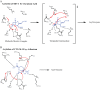
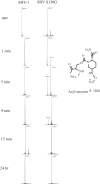
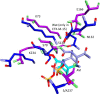
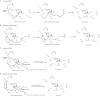
β-Lactamase enzymes (EC 3.5.2.6) are a significant threat to the continued use of β-lactam antibiotics to treat infections. A novel non-β-lactam β-lactamase inhibitor with activity against many class A and C and some class D β-lactamase variants, avibactam, is now available in the clinic in partnership with ceftazidime. Here, we explored the activity of avibactam against a variety of characterized isogenic laboratory constructs of β-lactamase inhibitor-resistant variants of the class A enzyme SHV (M69I/L/V, S130G, K234R, R244S, and N276D). We discovered that the S130G variant of SHV-1 shows the most significant resistance to inhibition by avibactam, based on both microbiological and biochemical characterizations. Using a constant concentration of 4 mg/liter of avibactam as a β-lactamase inhibitor in combination with ampicillin, the MIC increased from 1 mg/liter for blaSHV-1 to 256 mg/liter for blaSHV S130G expressed in Escherichia coli DH10B. At steady state, the k2/K value of the S130G variant when inactivated by avibactam was 1.3 M(-1) s(-1), versus 60,300 M(-1) s(-1) for the SHV-1 β-lactamase. Under timed inactivation conditions, we found that an approximately 1,700-fold-higher avibactam concentration was required to inhibit SHV S130G than the concentration that inhibited SHV-1. Molecular modeling suggested that the positioning of amino acids in the active site of SHV may result in an alternative pathway of inactivation when complexed with avibactam, compared to the structure of CTX-M-15-avibactam, and that S130 plays a role in the acylation of avibactam as a general acid/base. In addition, S130 may play a role in recyclization. As a result, we advance that the lack of a hydroxyl group at position 130 in the S130G variant of SHV-1 substantially slows carbamylation of the β-lactamase by avibactam by (i) removing an important proton acceptor and donator in catalysis and (ii) decreasing the number of H bonds. In addition, recyclization is most likely also slow due to the lack of a general base to initiate the process. Considering other inhibitor-resistant mechanisms among class A β-lactamases, S130 may be the most important amino acid for the inhibition of class A β-lactamases, perhaps even for the novel diazabicyclooctane class of β-lactamase inhibitors.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Variants of β-lactamase KPC-2 that are resistant to inhibition by avibactam.Antimicrob Agents Chemother. 2015 Jul;59(7):3710-7. doi: 10.1128/AAC.04406-14. Epub 2015 Feb 9. Antimicrob Agents Chemother. 2015. PMID: 25666153 Free PMC article.
-
Activity of ceftazidime/avibactam against isogenic strains of Escherichia coli containing KPC and SHV β-lactamases with single amino acid substitutions in the Ω-loop.J Antimicrob Chemother. 2015 Aug;70(8):2279-86. doi: 10.1093/jac/dkv094. Epub 2015 May 8. J Antimicrob Chemother. 2015. PMID: 25957381 Free PMC article.
-
Reclaiming the efficacy of β-lactam-β-lactamase inhibitor combinations: avibactam restores the susceptibility of CMY-2-producing Escherichia coli to ceftazidime.Antimicrob Agents Chemother. 2014 Aug;58(8):4290-7. doi: 10.1128/AAC.02625-14. Epub 2014 May 12. Antimicrob Agents Chemother. 2014. PMID: 24820081 Free PMC article.
-
Imipenem-Relebactam and Meropenem-Vaborbactam: Two Novel Carbapenem-β-Lactamase Inhibitor Combinations.Drugs. 2018 Jan;78(1):65-98. doi: 10.1007/s40265-017-0851-9. Drugs. 2018. PMID: 29230684 Review.
-
Focus on ceftazidime-avibactam for optimizing outcomes in complicated intra-abdominal and urinary tract infections.Expert Opin Investig Drugs. 2015;24(9):1261-73. doi: 10.1517/13543784.2015.1062873. Epub 2015 Jul 6. Expert Opin Investig Drugs. 2015. PMID: 26145447 Review.
Cited by
-
Identifying Oxacillinase-48 Carbapenemase Inhibitors Using DNA-Encoded Chemical Libraries.ACS Infect Dis. 2020 May 8;6(5):1214-1227. doi: 10.1021/acsinfecdis.0c00015. Epub 2020 Mar 25. ACS Infect Dis. 2020. PMID: 32182432 Free PMC article.
-
Resistance mechanisms and population structure of highly drug resistant Klebsiella in Pakistan during the introduction of the carbapenemase NDM-1.Sci Rep. 2019 Feb 20;9(1):2392. doi: 10.1038/s41598-019-38943-7. Sci Rep. 2019. PMID: 30787414 Free PMC article.
-
First Report of Ceftazidime-Avibactam Resistance in a KPC-3-Expressing Klebsiella pneumoniae Isolate.Antimicrob Agents Chemother. 2015 Oct;59(10):6605-7. doi: 10.1128/AAC.01165-15. Epub 2015 Jul 20. Antimicrob Agents Chemother. 2015. PMID: 26195508 Free PMC article.
-
Resistance to Ceftazidime/Avibactam, Meropenem/Vaborbactam and Imipenem/Relebactam in Gram-Negative MDR Bacilli: Molecular Mechanisms and Susceptibility Testing.Antibiotics (Basel). 2022 May 6;11(5):628. doi: 10.3390/antibiotics11050628. Antibiotics (Basel). 2022. PMID: 35625273 Free PMC article. Review.
-
Endless Resistance. Endless Antibiotics?Medchemcomm. 2016 Jan 1;7(1):37-49. doi: 10.1039/C5MD00394F. Epub 2015 Nov 3. Medchemcomm. 2016. PMID: 27746889 Free PMC article.
References
-
- Bush K, Palzkill T, Jacoby G. 2015. β-Lactamase classification and amino acid sequences for TEM, SHV and OXA extended-spectrum and inhibitor resistant enzymes. Lahey Clinic, Burlington, MA: http://www.lahey.org/Studies/ Accessed 1 July 2014.
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

