iASPP, a previously unidentified regulator of desmosomes, prevents arrhythmogenic right ventricular cardiomyopathy (ARVC)-induced sudden death
- PMID: 25691752
- PMCID: PMC4352828
- DOI: 10.1073/pnas.1408111112
iASPP, a previously unidentified regulator of desmosomes, prevents arrhythmogenic right ventricular cardiomyopathy (ARVC)-induced sudden death
Abstract
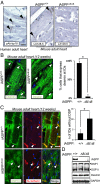
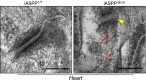
Desmosomes are anchoring junctions that exist in cells that endure physical stress such as cardiac myocytes. The importance of desmosomes in maintaining the homeostasis of the myocardium is underscored by frequent mutations of desmosome components found in human patients and animal models. Arrhythmogenic right ventricular cardiomyopathy (ARVC) is a phenotype caused by mutations in desmosomal components in ∼ 50% of patients, however, the causes in the remaining 50% of patients still remain unknown. A deficiency of inhibitor of apoptosis-stimulating protein of p53 (iASPP), an evolutionarily conserved inhibitor of p53, caused by spontaneous mutation recently has been associated with a lethal autosomal recessive cardiomyopathy in Poll Hereford calves and Wa3 mice. However, the molecular mechanisms that mediate this putative function of iASPP are completely unknown. Here, we show that iASPP is expressed at intercalated discs in human and mouse postmitotic cardiomyocytes. iASPP interacts with desmoplakin and desmin in cardiomyocytes to maintain the integrity of desmosomes and intermediate filament networks in vitro and in vivo. iASPP deficiency specifically induces right ventricular dilatation in mouse embryos at embryonic day 16.5. iASPP-deficient mice with exon 8 deletion (Ppp1r13l(Δ8/Δ8)) die of sudden cardiac death, displaying features of ARVC. Intercalated discs in cardiomyocytes from four of six human ARVC cases show reduced or loss of iASPP. ARVC-derived desmoplakin mutants DSP-1-V30M and DSP-1-S299R exhibit weaker binding to iASPP. These data demonstrate that by interacting with desmoplakin and desmin, iASPP is an important regulator of desmosomal function both in vitro and in vivo. This newly identified property of iASPP may provide new molecular insight into the pathogenesis of ARVC.
Keywords: ARVC; cell–cell junctions; desmosome; iASPP; sudden death.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Garrod D, Chidgey M. Desmosome structure, composition and function. Biochim Biophys Acta. 2008;1778(3):572–587. - PubMed
-
- Protonotarios N, Tsatsopoulou A. Naxos disease and Carvajal syndrome: Cardiocutaneous disorders that highlight the pathogenesis and broaden the spectrum of arrhythmogenic right ventricular cardiomyopathy. Cardiovasc Pathol. 2004;13(4):185–194. - PubMed
-
- Corrado D, et al. Spectrum of clinicopathologic manifestations of arrhythmogenic right ventricular cardiomyopathy/dysplasia: A multicenter study. J Am Coll Cardiol. 1997;30(6):1512–1520. - PubMed
-
- McRae AT, 3rd, Chung MK, Asher CR. Arrhythmogenic right ventricular cardiomyopathy: A cause of sudden death in young people. Cleve Clin J Med. 2001;68(5):459–467. - PubMed
-
- Lombardi R, Marian AJ. Molecular genetics and pathogenesis of arrhythmogenic right ventricular cardiomyopathy: A disease of cardiac stem cells. Pediatr Cardiol. 2011;32(3):360–365. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

