Patterns of hypothalamic regionalization in amphibians and reptiles: common traits revealed by a genoarchitectonic approach
- PMID: 25691860
- PMCID: PMC4315040
- DOI: 10.3389/fnana.2015.00003
Patterns of hypothalamic regionalization in amphibians and reptiles: common traits revealed by a genoarchitectonic approach
Abstract
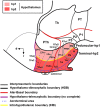
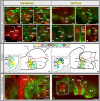
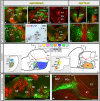
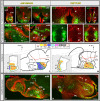
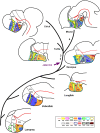
Most studies in mammals and birds have demonstrated common patterns of hypothalamic development highlighted by the combination of developmental regulatory genes (genoarchitecture), supporting the notion of the hypothalamus as a component of the secondary prosencephalon, topologically rostral to the diencephalon. In our comparative analysis we have summarized the data on the expression patterns of different transcription factors and neuroactive substances, used as anatomical markers, in the developing hypothalamus of the amphibian Xenopus laevis and the juvenile turtle Pseudemys scripta. This analysis served to highlight the organization of the hypothalamus in the anamniote/amniotic transition. We have identified supraoptoparaventricular and the suprachiasmatic regions (SCs) in the alar part of the hypothalamus, and tuberal and mammillary regions in the basal hypothalamus. Shared features in the two species are: (1) The supraoptoparaventricular region (SPV) is defined by the expression of Otp and the lack of Nkx2.1/Isl1. It is subdivided into rostral, rich in Otp and Nkx2.2, and caudal, only Otp-positive, portions. (2) The suprachiasmatic area contains catecholaminergic cell groups and lacks Otp, and can be further divided into rostral (rich in Nkx2.1 and Nkx2.2) and a caudal (rich in Isl1 and devoid of Nkx2.1) portions. (3) Expression of Nkx2.1 and Isl1 define the tuberal hypothalamus and only the rostral portion expresses Otp. (4) Its caudal boundary is evident by the lack of Isl1 in the adjacent mammillary region, which expresses Nkx2.1 and Otp. Differences in the anamnio-amniote transition were noted since in the turtle, like in other amniotes, the boundary between the alar hypothalamus and the telencephalic preoptic area shows distinct Nkx2.2 and Otp expressions but not in the amphibian (anamniote), and the alar SPV is defined by the expression of Otp/Pax6, whereas in Xenopus only Otp is expressed.
Keywords: development; evolution; forebrain patterning; hypothalamus; prosencephalon.
Figures


Similar articles
-
Subdivisions of the turtle Pseudemys scripta hypothalamus based on the expression of regulatory genes and neuronal markers.J Comp Neurol. 2012 Feb 15;520(3):453-78. doi: 10.1002/cne.22762. J Comp Neurol. 2012. PMID: 21935937
-
Characterization of the hypothalamus of Xenopus laevis during development. II. The basal regions.J Comp Neurol. 2014 Apr 1;522(5):1102-31. doi: 10.1002/cne.23471. J Comp Neurol. 2014. PMID: 24122702
-
Analysis of Islet-1, Nkx2.1, Pax6, and Orthopedia in the forebrain of the sturgeon Acipenser ruthenus identifies conserved prosomeric characteristics.J Comp Neurol. 2022 Apr;530(5):834-855. doi: 10.1002/cne.25249. Epub 2021 Oct 18. J Comp Neurol. 2022. PMID: 34547112
-
Contribution of genoarchitecture to understanding forebrain evolution and development, with particular emphasis on the amygdala.Brain Behav Evol. 2011;78(3):216-36. doi: 10.1159/000330056. Epub 2011 Aug 23. Brain Behav Evol. 2011. PMID: 21860224 Review.
-
The olfactory amygdala in amniotes: an evo-devo approach.Anat Rec (Hoboken). 2013 Sep;296(9):1317-32. doi: 10.1002/ar.22744. Epub 2013 Jul 31. Anat Rec (Hoboken). 2013. PMID: 23904411 Review.
Cited by
-
The Shark Alar Hypothalamus: Molecular Characterization of Prosomeric Subdivisions and Evolutionary Trends.Front Neuroanat. 2016 Nov 24;10:113. doi: 10.3389/fnana.2016.00113. eCollection 2016. Front Neuroanat. 2016. PMID: 27932958 Free PMC article.
-
Whole-Body Single-Cell Sequencing Reveals Transcriptional Domains in the Annelid Larval Body.Mol Biol Evol. 2018 May 1;35(5):1047-1062. doi: 10.1093/molbev/msx336. Mol Biol Evol. 2018. PMID: 29373712 Free PMC article.
-
The Shark Basal Hypothalamus: Molecular Prosomeric Subdivisions and Evolutionary Trends.Front Neuroanat. 2018 Mar 14;12:17. doi: 10.3389/fnana.2018.00017. eCollection 2018. Front Neuroanat. 2018. PMID: 29593505 Free PMC article.
-
Neuroimaging and immunofluorescence of the Pseudopus apodus brain: unraveling its structural complexity.Brain Struct Funct. 2025 May 28;230(5):76. doi: 10.1007/s00429-025-02940-6. Brain Struct Funct. 2025. PMID: 40434439 Free PMC article.
-
Xenopus leads the way: Frogs as a pioneering model to understand the human brain.Genesis. 2021 Feb;59(1-2):e23405. doi: 10.1002/dvg.23405. Epub 2020 Dec 27. Genesis. 2021. PMID: 33369095 Free PMC article. Review.
References
-
- Acampora D., Postiglione M. P., Avantaggiato V., Di Bonito M., Vaccarino F. M., Michaud J., et al. . (1999). Progressive impairment of developing neuroendocrine cell lineages in the hypothalamus of mice lacking the Orthopedia gene. Genes Dev. 13, 2787–2800. 10.1101/gad.13.21.2787 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

