Tolerability of High-Volume Subcutaneous Injections of a Viscous Placebo Buffer: A Randomized, Crossover Study in Healthy Subjects
- PMID: 25693652
- PMCID: PMC4674646
- DOI: 10.1208/s12249-015-0288-y
Tolerability of High-Volume Subcutaneous Injections of a Viscous Placebo Buffer: A Randomized, Crossover Study in Healthy Subjects
Erratum in
-
Erratum to: Tolerability of High-Volume Subcutaneous Injections of a Viscous Placebo Buffer: A Randomized, Crossover Study in Healthy Subjects.AAPS PharmSciTech. 2015 Dec;16(6):1500. doi: 10.1208/s12249-015-0324-y. AAPS PharmSciTech. 2015. PMID: 25904027 Free PMC article. No abstract available.
Abstract
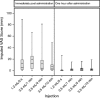
Monoclonal antibody biotherapeutics are often administered by subcutaneous (SC) injection. Due to dose requirements and formulation limitations, SC injections >1 mL are often required. We used a viscous placebo buffer (5 cP), characteristic of a high-concentration antibody formulation, to investigate the effect of dose volume and injection rate on the tolerability of higher-volume SC injections. In this randomized, crossover, single-center study, 48 healthy adults received one 1.2-mL bolus injection over 5 s and three 3.5-mL injections over 1, 4, and 10 min in different abdominal quadrants, with each injection separated by approximately 2 h. The primary objective was to compare pain scores associated with the injections, immediately after administration and 1 h later, using a 100-mm visual analog scale (VAS). Secondary objectives included assessment of adverse events, including injection site reactions and swelling. Mean age was 38.4 (11.6) years and 20 subjects (42%) were female. Lowest mean VAS score was for the 10-min (6.83 mm) and highest for the 1-min injection (19.13 mm). One hour after administration, mean VAS scores were <3.5 mm for all injections. Swelling was similar among the three 3.5-mL injections. After needle removal, leakage occurred following 14 (29%) 1.2-mL injections, eight (17%) 4-min injections, five (10%) 1-min injections, and four (8%) 10-min injections. Fifteen subjects (31%) experienced an adverse event, none of which was serious, fatal, or led to study discontinuation. All injection durations were well tolerated, suggesting a single large-volume SC injection of a biotherapeutic agent could be used instead of multiple injections.
Keywords: placebo buffer; subcutaneous injections; tolerability; viscous.
Figures
References
-
- Anderson G, Meyer D, Herrman CE, Sheppard C, Murray R, Fox EJ, et al. Tolerability and safety of novel half milliliter formulation of glatiramer acetate for subcutaneous injection: an open-label, multicenter, randomized comparative study. J Neurol. 2010;257(11):1917–23. doi: 10.1007/s00415-010-5779-x. - DOI - PMC - PubMed
-
- Davies M, Heller S, Sreenan S, Sapin H, Adetunji O, Tahbaz A, et al. Once-weekly exenatide versus once- or twice-daily insulin detemir: randomized, open-label, clinical trial of efficacy and safety in patients with type 2 diabetes treated with metformin alone or in combination with sulfonylureas. Diabetes Care. 2013;36(5):1368–76. doi: 10.2337/dc12-1333. - DOI - PMC - PubMed
-
- Giugliano RP, Desai NR, Kohli P, Rogers WJ, Somaratne R, Huang F, et al. Efficacy, safety, and tolerability of a monoclonal antibody to proprotein convertase subtilisin/kexin type 9 in combination with a statin in patients with hypercholesterolaemia (LAPLACE-TIMI 57): a randomised, placebo-controlled, dose-ranging, phase 2 study. Lancet. 2012;380(9858):2007–17. doi: 10.1016/S0140-6736(12)61770-X. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous