A1M/α1-microglobulin is proteolytically activated by myeloperoxidase, binds its heme group and inhibits low density lipoprotein oxidation
- PMID: 25698971
- PMCID: PMC4315848
- DOI: 10.3389/fphys.2015.00011
A1M/α1-microglobulin is proteolytically activated by myeloperoxidase, binds its heme group and inhibits low density lipoprotein oxidation
Abstract
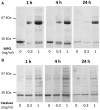
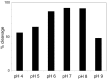
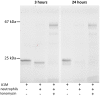
α1-microglobulin (A1M) is a 26 kDa plasma and tissue protein with reductase activity and radical- and heme-binding anti-oxidative functions. In addition, exposure of A1M to hemoglobin has been shown to induce proteolytic elimination of a C-terminal tetrapeptide yielding a heme-degrading form, truncated A1M (t-A1M). Myeloperoxidase (MPO), a heme-containing enzyme that catalyzes the production of free radicals and hypochlorite, is released by neutrophils during the inflammatory response to bacterial infections. MPO-induced low density lipoprotein (LDL)-oxidation in blood has been suggested as a causative factor in atherosclerosis. In this study we have hypothesized that A1M interacts with MPO in a similar mode as with hemoglobin, and is a regulator of its activity. The results show that A1M is proteolytically cleaved, with formation of t-A1M, after exposure to MPO, and that t-A1M contains iron and heme-degradation products. The reaction is dependent of pH, time and concentration of substrates and a pH-value around 7 is shown to be optimal for cleavage. Furthermore, A1M inhibits MPO- and hydrogen peroxide-induced oxidation of LDL. The results suggest that A1M may have a role as an inhibitor of the damaging effects of the neutrophil respiratory burst on bystander tissue components.
Keywords: C-terminal proteolysis; heme binding; low density lipoprotein; myeloperoxidase; neutrophils; α1-microglobulin.
Figures





Similar articles
-
Ferryl Hemoglobin and Heme Induce A1-Microglobulin in Hemorrhaged Atherosclerotic Lesions with Inhibitory Function against Hemoglobin and Lipid Oxidation.Int J Mol Sci. 2021 Jun 22;22(13):6668. doi: 10.3390/ijms22136668. Int J Mol Sci. 2021. PMID: 34206377 Free PMC article.
-
α1-Microglobulin Protects Against Bleeding-Induced Oxidative Damage in Knee Arthropathies.Front Physiol. 2018 Nov 16;9:1596. doi: 10.3389/fphys.2018.01596. eCollection 2018. Front Physiol. 2018. PMID: 30505280 Free PMC article.
-
α1-Microglobulin (A1M) Protects Human Proximal Tubule Epithelial Cells from Heme-Induced Damage In Vitro.Int J Mol Sci. 2020 Aug 13;21(16):5825. doi: 10.3390/ijms21165825. Int J Mol Sci. 2020. PMID: 32823731 Free PMC article.
-
Structure, Functions, and Physiological Roles of the Lipocalin α1-Microglobulin (A1M).Front Physiol. 2021 Mar 3;12:645650. doi: 10.3389/fphys.2021.645650. eCollection 2021. Front Physiol. 2021. PMID: 33746781 Free PMC article. Review.
-
A1M, an extravascular tissue cleaning and housekeeping protein.Free Radic Biol Med. 2014 Sep;74:274-82. doi: 10.1016/j.freeradbiomed.2014.06.025. Epub 2014 Jul 14. Free Radic Biol Med. 2014. PMID: 25035076 Review.
Cited by
-
Cardiac Hepatopathy: New Perspectives on Old Problems through a Prism of Endogenous Metabolic Regulations by Hepatokines.Antioxidants (Basel). 2023 Feb 17;12(2):516. doi: 10.3390/antiox12020516. Antioxidants (Basel). 2023. PMID: 36830074 Free PMC article. Review.
-
The Role of α1-Microglobulin (A1M) in Erythropoiesis and Erythrocyte Homeostasis-Therapeutic Opportunities in Hemolytic Conditions.Int J Mol Sci. 2020 Sep 30;21(19):7234. doi: 10.3390/ijms21197234. Int J Mol Sci. 2020. PMID: 33008134 Free PMC article. Review.
-
Emerging Evidence Linking the Liver to the Cardiovascular System: Liver-derived Secretory Factors.J Clin Transl Hepatol. 2023 Oct 28;11(5):1246-1255. doi: 10.14218/JCTH.2022.00122. Epub 2023 May 12. J Clin Transl Hepatol. 2023. PMID: 37577236 Free PMC article. Review.
-
Transcriptomic Modulation Reveals the Specific Cellular Response in Chinese Sea Bass (Lateolabrax maculatus) Gills under Salinity Change and Alkalinity Stress.Int J Mol Sci. 2023 Mar 20;24(6):5877. doi: 10.3390/ijms24065877. Int J Mol Sci. 2023. PMID: 36982950 Free PMC article.
-
Binding of the human antioxidation protein α1-microglobulin (A1M) to heparin and heparan sulfate. Mapping of binding site, molecular and functional characterization, and co-localization in vivo and in vitro.Redox Biol. 2021 May;41:101892. doi: 10.1016/j.redox.2021.101892. Epub 2021 Feb 10. Redox Biol. 2021. PMID: 33607500 Free PMC article.
References
-
- Åkerström B., Borregaard N., Flower D. R., Salie J. P. (eds). (2006). Lipocalins: An Introduction. Georgetown, TX: Landes Bioscience.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

