Randomised clinical trial: safety, tolerability, pharmacokinetics and pharmacodynamics of repeated doses of TAK-438 (vonoprazan), a novel potassium-competitive acid blocker, in healthy male subjects
- PMID: 25707624
- PMCID: PMC4654261
- DOI: 10.1111/apt.13121
Randomised clinical trial: safety, tolerability, pharmacokinetics and pharmacodynamics of repeated doses of TAK-438 (vonoprazan), a novel potassium-competitive acid blocker, in healthy male subjects
Abstract
Background: TAK-438 (vonoprazan) is a potassium-competitive acid blocker that reversibly inhibits gastric H(+) , K(+) -ATPase.
Aim: To evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of TAK-438 in healthy Japanese and non-Japanese men.
Methods: In two Phase I, randomised, double-blind, placebo-controlled studies, healthy men (Japan N = 60; UK N = 48) received TAK-438 10-40 mg once daily at a fixed dose level for 7 consecutive days. Assessments included safety, tolerability, pharmacokinetics and pharmacodynamics (intragastric pH).
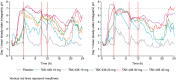
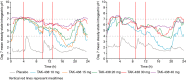
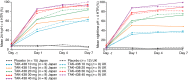
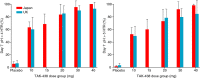
Results: Plasma concentration-time profiles of TAK-438 at all dose levels showed rapid absorption (median Tmax ≤2 h). Mean elimination half-life was up to 9 h. Exposure was slightly greater than dose proportional, with no apparent time-dependent inhibition of metabolism. There was no important difference between the two studies in AUC0-tau on Day 7. TAK-438 caused dose-dependent acid suppression. On Day 7, mean 24-h intragastric pH>4 holding time ratio (HTR) with 40 mg TAK-438 was 100% (Japan) and 93.2% (UK), and mean night-time pH>4 HTR was 100% (Japan) and 90.4% (UK). TAK-438 was well tolerated. The frequency of adverse events was similar at all dose levels and there were no serious adverse events. There were no important increases in serum alanine transaminase activity. Serum gastrin and pepsinogen I and II concentrations increased with TAK-438 dose.
Conclusions: TAK-438 in multiple rising oral dose levels of 10-40 mg once daily for 7 days was safe and well tolerated in healthy men and caused rapid, profound and sustained suppression of gastric acid secretion throughout each 24-h dosing interval. Clinicaltrials.gov identifiers: NCT02123953 and NCT02141711.
© 2015 The Authors. Alimentary Pharmacology and Therapeutics published by John Wiley & Sons Ltd.
Figures

Similar articles
-
Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of Single Rising TAK-438 (Vonoprazan) Doses in Healthy Male Japanese/non-Japanese Subjects.Clin Transl Gastroenterol. 2015 Jun 25;6(6):e94. doi: 10.1038/ctg.2015.18. Clin Transl Gastroenterol. 2015. PMID: 26111126 Free PMC article.
-
The First-in-Class Potassium-Competitive Acid Blocker, Vonoprazan Fumarate: Pharmacokinetic and Pharmacodynamic Considerations.Clin Pharmacokinet. 2016 Apr;55(4):409-18. doi: 10.1007/s40262-015-0326-7. Clin Pharmacokinet. 2016. PMID: 26369775 Review.
-
Safety, tolerability, pharmacodynamics and pharmacokinetics of DWP14012, a novel potassium-competitive acid blocker, in healthy male subjects.Aliment Pharmacol Ther. 2018 Jul;48(2):206-218. doi: 10.1111/apt.14818. Epub 2018 Jun 4. Aliment Pharmacol Ther. 2018. PMID: 29863280 Clinical Trial.
-
Characteristics of the Novel Potassium-Competitive Acid Blocker Vonoprazan Fumarate (TAK-438).Adv Ther. 2016 Jul;33(7):1140-57. doi: 10.1007/s12325-016-0345-2. Epub 2016 Jun 10. Adv Ther. 2016. PMID: 27287852 Review.
-
Pharmacokinetics and Safety of Triple Therapy with Vonoprazan, Amoxicillin, and Clarithromycin or Metronidazole: A Phase 1, Open-Label, Randomized, Crossover Study.Adv Ther. 2016 Sep;33(9):1519-35. doi: 10.1007/s12325-016-0374-x. Epub 2016 Jul 18. Adv Ther. 2016. PMID: 27432383 Clinical Trial.
Cited by
-
The Physiology of the Gastric Parietal Cell.Physiol Rev. 2020 Apr 1;100(2):573-602. doi: 10.1152/physrev.00016.2019. Epub 2019 Oct 31. Physiol Rev. 2020. PMID: 31670611 Free PMC article. Review.
-
Cytochrome P450-Based Drug-Drug Interactions of Vonoprazan In Vitro and In Vivo.Front Pharmacol. 2020 Feb 14;11:53. doi: 10.3389/fphar.2020.00053. eCollection 2020. Front Pharmacol. 2020. PMID: 32116727 Free PMC article.
-
The Effect of Food on the Pharmacokinetics of the Potassium-Competitive Acid Blocker Vonoprazan.Clin Pharmacol Drug Dev. 2022 Feb;11(2):278-284. doi: 10.1002/cpdd.1009. Epub 2021 Aug 24. Clin Pharmacol Drug Dev. 2022. PMID: 34431240 Free PMC article. Clinical Trial.
-
Potassium-Competitive Acid Blockers (P-CABs): Are They Finally Ready for Prime Time in Acid-Related Disease?Clin Transl Gastroenterol. 2015 Oct 29;6(10):e119. doi: 10.1038/ctg.2015.39. Clin Transl Gastroenterol. 2015. PMID: 26513137 Free PMC article.
-
Vonoprazan-based therapy versus standard regimen for Helicobacter pylori infection management in Egypt: an open-label randomized controlled trial.Sci Rep. 2025 May 8;15(1):15989. doi: 10.1038/s41598-025-98606-8. Sci Rep. 2025. PMID: 40341536 Free PMC article. Clinical Trial.
References
-
- Modlin IM, Sachs G. Acid Related Diseases: Biology and Treatment. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
