The effects of nanoparticle drug loading on the pharmacokinetics of anticancer agents
- PMID: 25707978
- PMCID: PMC4594874
- DOI: 10.2217/nnm.14.179
The effects of nanoparticle drug loading on the pharmacokinetics of anticancer agents
Abstract
Major advances in carrier-mediated agents, which include nanoparticles, nanosomes and conjugates, have revolutionized drug delivery capabilities over the past decade. While providing numerous advantages, such as greater solubility, duration of exposure and delivery to the site of action over their small-molecule counterparts, there is substantial variability in systemic clearance and distribution, tumor delivery and pharmacologic effects (efficacy and toxicity) of these agents. This review provides an overview of factors that affect the pharmacokinetics and pharmacodynamics of carrier-mediated agents in preclinical models and patients.
Keywords: Doxil; S-CKD602; carrier-mediated agents; clearance; nanoparticles; pharmacokinetics.
Figures

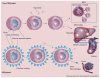
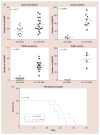
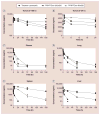
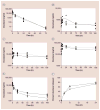

Similar articles
-
Complex effects of tumor microenvironment on the tumor disposition of carrier-mediated agents.Nanomedicine (Lond). 2017 Aug;12(16):2021-2042. doi: 10.2217/nnm-2017-0101. Epub 2017 Jul 26. Nanomedicine (Lond). 2017. PMID: 28745129 Free PMC article. Review.
-
Formulation and physiologic factors affecting the pharmacology of carrier-mediated anticancer agents.Expert Opin Drug Metab Toxicol. 2015;11(9):1419-33. doi: 10.1517/17425255.2015.1057496. Epub 2015 Jul 20. Expert Opin Drug Metab Toxicol. 2015. PMID: 26173794 Review.
-
Methods and Study Designs for Characterizing the Pharmacokinetics and Pharmacodynamics of Carrier-Mediated Agents.Methods Mol Biol. 2018;1831:201-228. doi: 10.1007/978-1-4939-8661-3_15. Methods Mol Biol. 2018. PMID: 30051434
-
Interpatient pharmacokinetic and pharmacodynamic variability of carrier-mediated anticancer agents.Clin Pharmacol Ther. 2012 May;91(5):802-12. doi: 10.1038/clpt.2012.12. Clin Pharmacol Ther. 2012. PMID: 22472987 Review.
-
Pharmacokinetics and efficacy of doxorubicin-loaded plant virus nanoparticles in preclinical models of cancer.Nanomedicine (Lond). 2017 Oct;12(20):2519-2532. doi: 10.2217/nnm-2016-0421. Epub 2017 Sep 27. Nanomedicine (Lond). 2017. PMID: 28952882
Cited by
-
Complex effects of tumor microenvironment on the tumor disposition of carrier-mediated agents.Nanomedicine (Lond). 2017 Aug;12(16):2021-2042. doi: 10.2217/nnm-2017-0101. Epub 2017 Jul 26. Nanomedicine (Lond). 2017. PMID: 28745129 Free PMC article. Review.
-
Immune response effects of diverse vaccine antigen attachment ways based on the self-made nanoemulsion adjuvant in systemic MRSA infection.RSC Adv. 2018 Mar 14;8(19):10425-10436. doi: 10.1039/c8ra00154e. eCollection 2018 Mar 13. RSC Adv. 2018. PMID: 35540467 Free PMC article.
-
Employing bicontinuous-to-micellar transitions in nanostructure morphology for on-demand photo-oxidation responsive cytosolic delivery and off-on cytotoxicity.Nanoscale. 2020 Mar 5;12(9):5332-5340. doi: 10.1039/c9nr10921h. Nanoscale. 2020. PMID: 32090217 Free PMC article.
-
Molecular Dynamics Studies of Poly(Lactic Acid) Nanoparticles and Their Interactions with Vitamin E and TLR Agonists Pam1CSK4 and Pam3CSK4.Nanomaterials (Basel). 2020 Nov 5;10(11):2209. doi: 10.3390/nano10112209. Nanomaterials (Basel). 2020. PMID: 33167538 Free PMC article.
-
Quality-by-Design Concepts to Improve Nanotechnology-Based Drug Development.Pharm Res. 2019 Sep 3;36(11):153. doi: 10.1007/s11095-019-2692-6. Pharm Res. 2019. PMID: 31482243 Review.
References
-
- Ge Y, Tiwari A, Li S. Nanomedicine – bridging the gap between nanotechnology and medicine. Adv Mat Lett. 2011;2(1):1–2.
-
- Caron WP, Song G, Kumar P, Rawal S, Zamboni WC. Interpatient pharmacokinetic and pharmacodynamic variability of carrier-mediated anticancer agents. Clin Pharmacol Ther. 2012;91(5):802–812. - PubMed
-
- Laginha K, Mumbengegwi D, Allen T. Liposomes targeted via two different antibodies: assay, B-cell binding and cytotoxicity. Biochim Biophys Acta. 2005;1711(1):25–32. - PubMed
-
- Zamboni WC. Liposomal, nanoparticle, and conjugated formulations of anticancer agents. Clin Cancer Res. 2005;11(23):8230–8234. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
